Neuroimaging of the Wernicke-Korsakoff syndrome
- PMID: 19066199
- PMCID: PMC2724861
- DOI: 10.1093/alcalc/agn103
Neuroimaging of the Wernicke-Korsakoff syndrome
Abstract
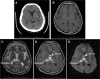
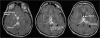
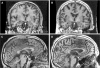
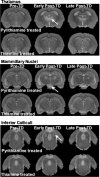
Aim: Presented is the neuroradiological signature of acute Wernicke's encephalopathy (WE), derived from different types of magnetic resonance imaging (MRI) sequences. WE results from thiamine depletion, and its most typical antecedent is chronic alcohol dependence. Brain regions observed with in vivo MRI affected in acute WE include the mammillary bodies, periaqueductal and periventricular gray matter, collicular bodies and thalamus. These affected areas are usually edematous and are best visualized and quantified with MRI sequences that highlight such tissue. Following the acute WE phase and resolution of edema and inflammation of affected brain tissue, WE, if not adequately treated with thiamine repletion, can herald Korsakoff's syndrome (KS), with its symptomatic hallmark of global amnesia, that is, the inability to commit newly encountered (episodic) information to memory for later recall or recognition.
Methods: Neuropathology of KS detectable with MRI has a different neuroradiological signature from the acute stage and can be observed as tissue shrinkage or atrophy of selective brain structures, including the mammillary bodies and thalamus and ventricular expansion, probably indicative of atrophy of surrounding gray matter nuclei. Quantification of these and additional gray matter structures known to underlie global amnesia reveal substantial bilateral volume deficits in the hippocampus, in addition to the mammillary bodies and thalamus, and modest deficits in the medial septum/diagonal band of Broca. The infratentorium is also affected, exhibiting volume deficits in cerebellar hemispheres, anterior superior vermis and pons, contributing to ataxia of gait and stance.
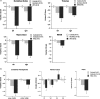
Results: Consideration of WKS structural brain changes in the context of the neuropathology of non-WKS alcoholism revealed a graded pattern of volume deficits, from mild in non-WKS alcoholics to moderate or severe in WKS, in the mammillary bodies, hippocampus, thalamus, cerebellum and pons. The development and resolution of brain structures affected in acute, chronic and treated WE was verified in longitudinal MRI study of rats that modeled of the interaction of extensive alcohol consumption and thiamine depletion and repletion.
Conclusions: Thus, neuroradiological examination with MRI is valuable in the diagnosis of acute WE and enables in vivo tracking of the progression of the brain pathology of WE from the acute pathological phase to resolution with thiamine treatment or to progression to KS without treatment. Further, in vivo MRI facilitates translational studies to model antecedent conditions contributing to the development, sequelae and treatment of WE.
Figures

References
-
- Antunez E, Estruch R, Cardenal C, et al. Usefulness of CT and MR imaging in the diagnosis of acute Wernicke's encephalopathy. AJR Am J Roentgenol. 1998;171:1131–7. - PubMed
-
- Ashikaga R, Araki Y, Ono Y, et al. FLAIR appearance of Wernicke encephalopathy. Radiat Med. 1997;15:251–3. - PubMed
-
- Baker K, Harding A, Halliday G, et al. Neuronal loss in functional zones of the cerebellum of chronic alcoholics with and without Wernicke's encephalopathy. Neuroscience. 1999;91:429–38. - PubMed

