Novel acridine-based compounds that exhibit an anti-pancreatic cancer activity are catalytic inhibitors of human topoisomerase II
- PMID: 19071108
- PMCID: PMC2637346
- DOI: 10.1016/j.ejphar.2008.11.044
Novel acridine-based compounds that exhibit an anti-pancreatic cancer activity are catalytic inhibitors of human topoisomerase II
Abstract
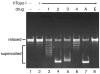
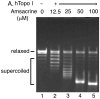
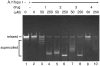
We have identified a small library of novel substituted 9-aminoacridine derivatives that inhibit cell proliferation of pancreatic cancer cell lines by inducing apoptosis [Goodell, J.R. et al., 2008. J. Med. Chem. 51, 179-182.]. To further investigate their antiproliferative activities, we have assessed the antiproliferative activity of these acridine-based compounds against several pancreatic cancer cell lines. All four compounds used in this study inhibited the proliferation of pancreatic cancer cell lines in vitro. In addition, we have employed a xenograft tumor model and found that these compounds also inhibit the proliferation of pancreatic cancer in vivo. In light of the potential importance of the anticancer activity of these acridine-based compounds, we have conducted a series of biochemical assays to determine the effect of these compounds on human topoisomerase II. Unlike amsacrine, these compounds do not poison topoisomerase II. Similar to amsacrine, however, these compounds intercalate into DNA in a way that they would alter the apparent topology of the DNA substrate. Thus, inhibition of the relaxation activity of topoisomerase II by these compounds has been reexamined using a DNA strand passage assay. We have found that these compounds, indeed, inhibit the catalytic activity of topoisomerase II. Thus, these novel acridine-based compounds with anti-pancreatic cancer activity are catalytic inhibitors, not poisons, of human topoisomerase II.
Figures
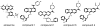





Similar articles
-
Novel tetra-acridine derivatives as dual inhibitors of topoisomerase II and the human proteasome.Biochem Pharmacol. 2007 Jun 15;73(12):1863-72. doi: 10.1016/j.bcp.2007.02.016. Epub 2007 Mar 3. Biochem Pharmacol. 2007. PMID: 17391647
-
Novel trifluoromethylated 9-amino-3,4-dihydroacridin-1(2H)-ones act as covalent poisons of human topoisomerase IIα.Bioorg Med Chem Lett. 2017 Feb 1;27(3):586-589. doi: 10.1016/j.bmcl.2016.12.011. Epub 2016 Dec 5. Bioorg Med Chem Lett. 2017. PMID: 27998679 Free PMC article.
-
Targeting Topoisomerase II Activity in NSCLC with 9-Aminoacridine Derivatives.Anticancer Res. 2015 Oct;35(10):5211-7. Anticancer Res. 2015. PMID: 26408679
-
A Review on Acridines as Antiproliferative Agents.Mini Rev Med Chem. 2022;22(21):2769-2798. doi: 10.2174/1389557522666220511125744. Mini Rev Med Chem. 2022. PMID: 35546777 Review.
-
Acridine derivatives as inhibitors/poisons of topoisomerase II.J Appl Toxicol. 2022 Apr;42(4):544-552. doi: 10.1002/jat.4238. Epub 2021 Sep 12. J Appl Toxicol. 2022. PMID: 34514603 Review.
Cited by
-
Biological assessment of new tetrahydroacridine derivatives with fluorobenzoic moiety in vitro on A549 and HT-29 cell lines and in vivo on animal model.Hum Cell. 2020 Jul;33(3):859-867. doi: 10.1007/s13577-020-00376-0. Epub 2020 May 24. Hum Cell. 2020. PMID: 32449113 Free PMC article.
-
Microwave-assisted improved synthesis of pyrrolo[2,3,4-kl]acridine and dihydropyrrolo[2,3,4-kl]acridine derivatives catalyzed by silica sulfuric acid.Molecules. 2013 Jan 28;18(2):1613-25. doi: 10.3390/molecules18021613. Molecules. 2013. PMID: 23358320 Free PMC article.
-
Riccardin D, a novel macrocyclic bisbibenzyl, induces apoptosis of human leukemia cells by targeting DNA topoisomerase II.Invest New Drugs. 2012 Feb;30(1):212-22. doi: 10.1007/s10637-010-9554-8. Epub 2010 Oct 6. Invest New Drugs. 2012. PMID: 20924640
-
Synthesis of acridines by the [4 + 2] annulation of arynes and 2-aminoaryl ketones.J Org Chem. 2010 Apr 2;75(7):2289-95. doi: 10.1021/jo1000687. J Org Chem. 2010. PMID: 20222700 Free PMC article.
-
9-Aminoacridine Inhibits Ribosome Biogenesis by Targeting Both Transcription and Processing of Ribosomal RNA.Int J Mol Sci. 2022 Jan 23;23(3):1260. doi: 10.3390/ijms23031260. Int J Mol Sci. 2022. PMID: 35163183 Free PMC article.
References
-
- Adjei AA, Charron M, Rowinsky EK, Svingen PA, Miller J, Reid JM, Sebolt-Leopold J, Ames MM, Kaufmann SH. Effect of pyrazoloacridine (NSC 366140) on DNA topoisomerases I and II. Clin Cancer Res. 1998;4:683–691. - PubMed
-
- Broeker PL, Super HG, Thirman MJ, Pomykala H, Yonebayashi Y, Tanabe S, Tanabe S, Zeleznik-Le N, Rowley JD. Distribution of 11q23 breakpoints within the MLL breakpoint cluster region in de novo acute leukemia and in treatment-related acute myeloid leukemia: correlation with scaffold attachment regions and topoisomerase II consensus binding sites. Blood. 1996;87:1912–1922. - PubMed
-
- Cao D, Maitra A, Saavedra JA, Klimstra DS, Adsay NV, Hruban RH. Expression of novel markers of pancreatic ductal adenocarcinoma in pancreatic nonductal neoplasms: additional evidence of different genetic pathways. Mod Pathol. 2005;18:752–761. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

