Interaction of monocarboxylate transporter 4 with beta1-integrin and its role in cell migration
- PMID: 19073896
- PMCID: PMC2660264
- DOI: 10.1152/ajpcell.00430.2008
Interaction of monocarboxylate transporter 4 with beta1-integrin and its role in cell migration
Abstract
Monocarboxylate transporter (MCT) 4 is a heteromeric proton-coupled lactate transporter that is noncovalently linked to the extracellular matrix metalloproteinase inducer CD147 and is typically expressed in glycolytic tissues. There is increasing evidence to suggest that ion transporters are part of macromolecular complexes involved in regulating beta(1)-integrin adhesion and cell movement. In the present study we examined whether MCTs play a role in cell migration through their interaction with beta(1)-integrin. Using reciprocal coimmunoprecipitation assays, we found that beta(1)-integrin selectively associated with MCT4 in ARPE-19 and MDCK cells, two epithelial cell lines that express both MCT1 and MCT4. In polarized monolayers of ARPE-19 cells, MCT4 and beta(1)-integrin colocalized to the basolateral membrane, while both proteins were found in the leading edge lamellapodia of migrating cells. In scratch-wound assays, MCT4 knockdown slowed migration and increased focal adhesion size. In contrast, silencing MCT1 did not alter the rate of cell migration or focal adhesion size. Taken together, our findings suggest that the specific interaction of MCT4 with beta(1)-integrin may regulate cell migration through modulation of focal adhesions.
Figures
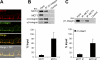

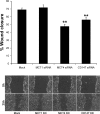
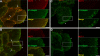

Similar articles
-
Polarized expression of monocarboxylate transporters in human retinal pigment epithelium and ARPE-19 cells.Invest Ophthalmol Vis Sci. 2003 Apr;44(4):1716-21. doi: 10.1167/iovs.02-0287. Invest Ophthalmol Vis Sci. 2003. PMID: 12657613
-
Monocarboxylate transporters (MCTs) in gliomas: expression and exploitation as therapeutic targets.Neuro Oncol. 2013 Feb;15(2):172-88. doi: 10.1093/neuonc/nos298. Epub 2012 Dec 20. Neuro Oncol. 2013. PMID: 23258846 Free PMC article.
-
CD147 regulates the expression of MCT1 and lactate export in multiple myeloma cells.Cell Cycle. 2013 Oct 1;12(19):3175-83. doi: 10.4161/cc.26193. Epub 2013 Sep 3. Cell Cycle. 2013. PMID: 24013424 Free PMC article.
-
Monocarboxylic acid transport.Compr Physiol. 2013 Oct;3(4):1611-43. doi: 10.1002/cphy.c130008. Compr Physiol. 2013. PMID: 24265240 Review.
-
The expression of lactate transporters (MCT1 and MCT4) in heart and muscle.Eur J Appl Physiol. 2001 Nov;86(1):6-11. doi: 10.1007/s004210100516. Eur J Appl Physiol. 2001. PMID: 11820324 Review.
Cited by
-
The clinicopathological significance of monocarboxylate transporters in testicular germ cell tumors.Oncotarget. 2018 Apr 17;9(29):20386-20398. doi: 10.18632/oncotarget.24910. eCollection 2018 Apr 17. Oncotarget. 2018. PMID: 29755659 Free PMC article.
-
Transport Metabolons and Acid/Base Balance in Tumor Cells.Cancers (Basel). 2020 Apr 7;12(4):899. doi: 10.3390/cancers12040899. Cancers (Basel). 2020. PMID: 32272695 Free PMC article. Review.
-
Clinical Significance of CD147 in Children with Inflammatory Bowel Disease.Biomed Res Int. 2020 Sep 15;2020:7647181. doi: 10.1155/2020/7647181. eCollection 2020. Biomed Res Int. 2020. PMID: 33015178 Free PMC article.
-
Identification of Prognostic Organic Cation and Anion Transporters in Different Cancer Entities by In Silico Analysis.Int J Mol Sci. 2020 Jun 24;21(12):4491. doi: 10.3390/ijms21124491. Int J Mol Sci. 2020. PMID: 32599841 Free PMC article.
-
NaBC1 Boron Transporter Enables Myoblast Response to Substrate Rigidity via Fibronectin-Binding Integrins.Adv Sci (Weinh). 2025 May;12(20):e2407548. doi: 10.1002/advs.202407548. Epub 2025 Apr 24. Adv Sci (Weinh). 2025. PMID: 40270477 Free PMC article.
References
-
- Ahmed N, Riley C, Rice G, Quinn M. Role of integrin receptors for fibronectin, collagen and laminin in the regulation of ovarian carcinoma functions in response to a matrix microenvironment. Clin Exp Metastasis 22: 391–402, 2005. - PubMed
-
- Berditchevski F, Chang S, Bodorova J, Hemler ME. Generation of monoclonal antibodies to integrin-associated proteins. Evidence that alpha3beta1 complexes with EMMPRIN/basigin/OX47/M6. J Biol Chem 272: 29174–29180, 1997. - PubMed
-
- Biswas C, Zhang Y, DeCastro R, Guo H, Nakamura T, Kataoka H, Nabeshima K. The human tumor cell-derived collagenase stimulatory factor (renamed EMMPRIN) is a member of the immunoglobulin superfamily. Cancer Res 55: 434–439, 1995. - PubMed
-
- Bourguignon LY, Singleton PA, Diedrich F, Stern R, Gilad E. CD44 interaction with Na+-H+ exchanger (NHE1) creates acidic microenvironments leading to hyaluronidase-2 and cathepsin B activation and breast tumor cell invasion. J Biol Chem 279: 26991–27007, 2004. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

