Interleukin-1 regulates the expression of sphingosine kinase 1 in glioblastoma cells
- PMID: 19074142
- PMCID: PMC2635028
- DOI: 10.1074/jbc.M807170200
Interleukin-1 regulates the expression of sphingosine kinase 1 in glioblastoma cells
Abstract
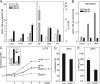
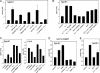
Chronic inflammation and inflammatory cytokines have recently been implicated in the development and progression of various types of cancer. In the brain, neuroinflammatory cytokines affect the growth and differentiation of both normal and malignant glial cells, with interleukin 1 (IL-1) shown to be secreted by the majority of glioblastoma cells. Recently, elevated levels of sphingosine kinase 1 (SphK1), but not SphK2, were correlated with a shorter survival prognosis for patients with glioblastoma multiforme. SphK1 is a lipid kinase that produces the pro-growth, anti-apoptotic sphingosine 1-phosphate, which can induce invasion of glioblastoma cells. Here, we show that the expression of IL-1 correlates with the expression of SphK1 in glioblastoma cells, and neutralizing anti-IL-1 antibodies inhibit both the growth and invasion of glioblastoma cells. Furthermore, IL-1 up-regulates SphK1 mRNA levels, protein expression, and activity in both primary human astrocytes and various glioblastoma cell lines; however, it does not affect SphK2 expression. The IL-1-induced SphK1 up-regulation can be blocked by the inhibition of JNK, the overexpression of the dominant-negative c-Jun(TAM67), and the down-regulation of c-Jun expression by small interference RNA. Activation of SphK1 expression by IL-1 occurs on the level of transcription and is mediated via a novel AP-1 element located within the first intron of the sphk1 gene. In summary, our results suggest that SphK1 expression is transcriptionally regulated by IL-1 in glioblastoma cells, and this pathway may be important in regulating survival and invasiveness of glioblastoma cells.
Figures



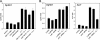

References
-
- Belda-Iniesta, C., de Castro Carpeno, J., Casado Saenz, E., Cejas Guerrero, P., Perona, R., and Gonzalez Baron, M. (2006) Clin. Transl. Oncol. 8 635–641 - PubMed
-
- Gilbertson, R. J., and Rich, J. N. (2007) Nat. Rev. Cancer 7 733–736 - PubMed
-
- Hill, C., Hunter, S. B., and Brat, D. J. (2003) Adv. Anat. Pathol. 10 212–217 - PubMed
-
- Deb, P., Sharma, M. C., Mahapatra, A. K., Agarwal, D., and Sarkar, C. (2005) Neurol. India 53 329–332 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

