ATP consumption by mammalian rod photoreceptors in darkness and in light
- PMID: 19084410
- PMCID: PMC2615811
- DOI: 10.1016/j.cub.2008.10.029
ATP consumption by mammalian rod photoreceptors in darkness and in light
Abstract
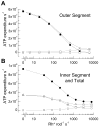
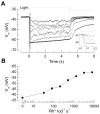
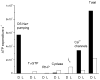
Why do vertebrates use rods and cones that hyperpolarize, when in insect eyes a single depolarizing photoreceptor can function at all light levels? We answer this question at least in part with a comprehensive assessment of ATP consumption for mammalian rods from voltages and currents and recently published physiological and biochemical data. In darkness, rods consume 10(8) ATP s(-1), about the same as Drosophila photoreceptors. Ion fluxes associated with phototransduction and synaptic transmission dominate; as in CNS, the contribution of enzymes of the second-messenger cascade is surprisingly small. Suppression of rod responses in daylight closes light-gated channels and reduces total energy consumption by >75%, but in Drosophila light opens channels and increases consumption 5-fold. Rods therefore provide an energy-efficient mechanism not present in rhabdomeric photoreceptors. Rods are metabolically less "costly" than cones, because cones do not saturate in bright light and use more ATP s(-1) for transducin activation and rhodopsin phosphorylation. This helps to explain why the vertebrate retina is duplex, and why some diurnal animals like primates have a small number of cones, concentrated in a region of high acuity.
Figures
References
-
- Hardie RC, Raghu P. Visual transduction in Drosophila. Nature. 2001;413:186–193. - PubMed
-
- Fain GL. Sensory Transduction. Sunderland, MA: Sinauer; 2003.
-
- Attwell D, Laughlin SB. An energy budget for signaling in the grey matter of the brain. Journal of Cerebral Blood Flow and Metabolism. 2001;21 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

