Effect of neuropeptide Y Y2 receptor deletion on emotional stress-induced neuronal activation in mice
- PMID: 19084906
- PMCID: PMC3034873
- DOI: 10.1002/syn.20597
Effect of neuropeptide Y Y2 receptor deletion on emotional stress-induced neuronal activation in mice
Abstract
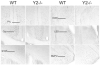
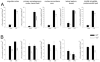
In different behavioral paradigms including the elevated plus maze (EPM), it was observed previously that deletion of the neuropeptide Y Y2 receptor subtype results in potent suppression of anxiety-related and stress-related behaviors. To identify neurobiological correlates underlying this behavioral reactivtiy, expression of c-Fos, an established early marker of neuronal activation, was examined in Y2 receptor knockout (Y2(-/-)) vs. wildtype (WT) mice. Mice were placed on the open arm (OA) or closed arm (CA) of the EPM for 10 min and the effect on regional c-Fos expression in the brain was investigated. The number of c-Fos positive neurons was significantly increased in both WT and Y2(-/-) lines after OA and CA exposure in 51 of 54 regions quantified. These regions included various cortical, limbic, thalamic, hypothalamic, and hindbrain regions. Genotype influenced c-Fos responses to arm exposures in 6 of the 51 activated regions: the cingulate cortex, barrel field of the primary somatosensory cortex, nucleus accumbens, dorsal lateral septum, amygdala and lateral periaqueductal gray. These differences in neuronal activity responses to the novel environments were more pronounced after OA than after CA exposure. Mice lacking Y2 receptors exhibited reduced neuronal activation when compared to WT animals in response to the emotional stressors. Reduced neuronal excitability in the identified brain areas relevant to the processing of motivated, explorative as well as anxiety-related behaviors is suggested to contribute to the reduced anxiety-related behavior observed in Y2(-/-) mice.
(c) 2008 Wiley-Liss, Inc.
Figures

Similar articles
-
Conditional CRF receptor 1 knockout mice show altered neuronal activation pattern to mild anxiogenic challenge.Psychopharmacology (Berl). 2006 Oct;188(3):374-85. doi: 10.1007/s00213-006-0513-1. Epub 2006 Sep 5. Psychopharmacology (Berl). 2006. PMID: 16953386
-
Increased novelty-induced motor activity and reduced depression-like behavior in neuropeptide Y (NPY)-Y4 receptor knockout mice.Neuroscience. 2009 Feb 18;158(4):1717-30. doi: 10.1016/j.neuroscience.2008.11.048. Epub 2008 Dec 9. Neuroscience. 2009. PMID: 19121371 Free PMC article.
-
Endogenous neuropeptide Y depresses the afferent signaling of gastric acid challenge to the mouse brainstem via neuropeptide Y type Y2 and Y4 receptors.Neuroscience. 2005;136(4):1097-107. doi: 10.1016/j.neuroscience.2005.08.038. Epub 2005 Oct 10. Neuroscience. 2005. PMID: 16216428 Free PMC article.
-
Neuropeptide Y-Y2 receptor knockout mice: influence of genetic background on anxiety-related behaviors.Neuroscience. 2011 Mar 10;176:420-30. doi: 10.1016/j.neuroscience.2010.10.075. Epub 2010 Nov 3. Neuroscience. 2011. PMID: 21055451
-
The role of Neuropeptide Y in fear conditioning and extinction.Neuropeptides. 2016 Feb;55:111-26. doi: 10.1016/j.npep.2015.09.007. Epub 2015 Sep 25. Neuropeptides. 2016. PMID: 26444585 Review.
Cited by
-
Magnesium deficiency induces anxiety and HPA axis dysregulation: modulation by therapeutic drug treatment.Neuropharmacology. 2012 Jan;62(1):304-12. doi: 10.1016/j.neuropharm.2011.07.027. Epub 2011 Aug 4. Neuropharmacology. 2012. PMID: 21835188 Free PMC article.
-
Rhythmic brain stimulation reduces anxiety-related behavior in a mouse model based on meditation training.Proc Natl Acad Sci U S A. 2017 Mar 7;114(10):2532-2537. doi: 10.1073/pnas.1700756114. Epub 2017 Feb 21. Proc Natl Acad Sci U S A. 2017. PMID: 28223484 Free PMC article.
-
Lactation reduces stress-caused dopaminergic activity and enhances GABAergic activity in the rat medial prefrontal cortex.J Mol Neurosci. 2014 Apr;52(4):515-24. doi: 10.1007/s12031-013-0104-7. Epub 2013 Oct 2. J Mol Neurosci. 2014. PMID: 24085524
-
Targeting the Neuropeptide Y System in Stress-related Psychiatric Disorders.Neurobiol Stress. 2015 Jan 1;1:33-43. doi: 10.1016/j.ynstr.2014.09.007. Neurobiol Stress. 2015. PMID: 25506604 Free PMC article.
-
Emerging neuropeptide targets in inflammation: NPY and VIP.Am J Physiol Gastrointest Liver Physiol. 2013 Jun 1;304(11):G949-57. doi: 10.1152/ajpgi.00493.2012. Epub 2013 Mar 28. Am J Physiol Gastrointest Liver Physiol. 2013. PMID: 23538492 Free PMC article. Review.
References
-
- Bacchi F, Mathe AA, Jimenez P, Stasi L, Arban R, Gerrard P, Caberlotto L. Anxiolytic-like effect of the selective neuropeptide Y Y2 receptor antagonist BIIE0246 in the elevated plus-maze. Peptides. 2006;27:3202–3207. - PubMed
-
- Charney DS, Grillon C, Bremner DJ. The neurobiological basis of anxiety and fear: Circuits, mechanisms, and neurochemical interactions (part I) Neuroscientist. 1998;4:35–44.
-
- Chronwall BM. Anatomy and physiology of the neuroendocrine arcuate nucleus. Peptides. 1985;6(Suppl 2):1–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
