Mechanism of metabolic activation and DNA adduct formation by the human carcinogen diethylstilbestrol: the defining link to natural estrogens
- PMID: 19089919
- PMCID: PMC2814601
- DOI: 10.1002/ijc.24113
Mechanism of metabolic activation and DNA adduct formation by the human carcinogen diethylstilbestrol: the defining link to natural estrogens
Abstract
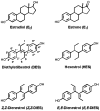
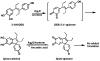
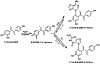
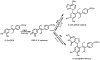
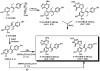
Diethylstilbestrol (DES) is a human carcinogen, based on sufficient epidemiological evidence. DES is mainly metabolized to its catechol, 3'-hydroxyDES (3'-OH-DES), which can further oxidize to DES-3',4'-quinone (DES-3',4'-Q). Similarly to estradiol-3,4-quinone, the reaction of DES-3',4'-Q with DNA would form the depurinating 3'-OH-DES-6'-N3Ade and 3'-OH-DES-6'-N7Gua adducts. To prove this hypothesis, synthesis of DES-3',4'-Q by oxidation of 3'-OH-DES with Ag(2)O was tried; this failed due to instantaneous formation of a spiro-quinone. Oxidation of 3'-OH-DES by lactoperoxidase or tyrosinase in the presence of DNA led to the formation of 3'-OH-DES-6'-N3Ade and 3'-OH-DES-6'-N7Gua adducts. These adducts were tentatively identified by LC-MS/MS as 3'-OH-DES-6'-N3Ade, m/z = 418 [M+H](+), and 3'-OH-DES-6'-N7Gua, m/z = 434 [M+H](+). Demonstration of their structures derived from their oxidation by MnO(2) to the DES quinone adducts and subsequent tautomerization to the dienestrol (DIES) catechol adducts, which are identical to the standard 3'-OH-DIES-6'-N3Ade, m/z = 416 [M+H](+), and 3'-OH-DIES-6'-N7Gua, m/z = 432 [M+H](+), adducts. The reaction of DIES-3',4'-Q or lactoperoxidase-activated 3'-OH-DIES with DNA did not produce any depurinating adducts, due to the dienic chain being perpendicular to the phenyl planes, which impedes the intercalation of DIES into the DNA. Enzymic oxidation of 3'-OH-DES suggests that the catechol of DES intercalates into DNA and is then oxidized to its quinone to yield N3Ade and N7Gua adducts. These results suggest that the common denominator of tumor initiation by the synthetic estrogen DES and the natural estrogen estradiol is formation of their catechol quinones, which react with DNA to afford the depurinating N3Ade and N7Gua adducts.
Figures
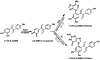

References
-
- Dodds EC, Goldberg L, Lawson W, Robinson R. Estrogenic activity of certain synthetic compounds. Nature. 1938;141:247–8.
-
- Herbst AL, Ulfelder H, Poskanzer DC. Adenocarcinoma of the vagina. Association of maternal stilbestrol therapy with tumor appearance in young women. N Engl J Med. 1971;284:878–81. - PubMed
-
- Swan SH. Diethylstilbestrol and clear cell vaginal carcinoma. Am J Med. 1987;83:372–4. - PubMed
-
- Noller KL, O’Brien PC. Diethylstilbestrol and clear cell vaginal carcinoma. Am J Med. 1987;83:374. - PubMed
-
- Herbst AL. Diethylstilbestrol and clear cell vaginal carcinoma. Am J Med. 1987;83:375–6. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

