A dual-signal regulatory circuit activates transcription of a set of divergent operons in Salmonella typhimurium
- PMID: 19091955
- PMCID: PMC2634881
- DOI: 10.1073/pnas.0807071106
A dual-signal regulatory circuit activates transcription of a set of divergent operons in Salmonella typhimurium
Abstract
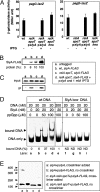
We present a molecular mechanism for signal transduction that activates transcription of the SlyA regulon in Salmonella typhimurium. We demonstrate that SlyA mediates transcriptional activation in response to guanosine tetraphosphate, ppGpp, according to the following observations: (i) in vivo transcription of SlyA-dependent genes is repressed when ppGpp is absent; this transcription can be restored by overproducing SlyA; (ii) in vivo dimerization and binding of SlyA to the target promoter are facilitated in the presence of ppGpp; and (iii) in vitro SlyA binding to the target promoter is enhanced when ppGpp is supplemented. Thus, ppGpp must be the cytoplasmic component that stimulates SlyA regulatory function by interacting directly with this regulator in Salmonella. This signaling domain, integrated by the PhoP/PhoQ 2-component system that activates slyA transcription by sensing Mg(2+), forms feedforward loops that regulate chromosomal loci identified through a motif search over the S. typhimurium genome. Many such loci are divergent operons, each formed by 2 neighboring genes in which transcription of these 2 loci proceeds in opposite directions. Both genes, however, are controlled by PhoP and SlyA through a single shared PhoP box and SlyA box present in their intergenic regions. A substitution in either box sequence causes a simultaneous cessation of transcription of a divergent operon, pagD-pagC, equivalent to the phenotype in a phoP or slyA mutant. We also identified several chromosomal loci that possess pagC-type genes without the cognate pagD-type genes. Therefore, our results provide a molecular basis for the understanding of SlyA-dependent phenotypes associated with Salmonella virulence.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Shi Y, Latifi T, Cromie M-J, Groisman E-A. Transcriptional control of the antimicrobial peptide resistance ugtL gene by the Salmonella PhoP and SlyA regulatory proteins. J Biol Chem. 2004;279:38618–38625. - PubMed
-
- Navarre W-W, et al. Co-regulation of Salmonella enterica genes required for virulence and resistance to antimicrobial peptides by SlyA and PhoP/PhoQ. Mol Microbiol. 2005;56:492–508. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

