Two PKC consensus sites on human acid-sensing ion channel 1b differentially regulate its function
- PMID: 19091960
- PMCID: PMC2643847
- DOI: 10.1152/ajpcell.00200.2008
Two PKC consensus sites on human acid-sensing ion channel 1b differentially regulate its function
Abstract
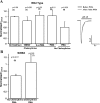
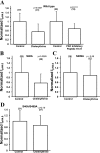
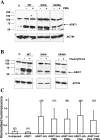
Human acid-sensing ion channel 1b (hASIC1b) is a H(+)-gated amiloride-sensitive cation channel. We have previously shown that glioma cells exhibit an amiloride-sensitive cation conductance. Amiloride and the ASIC1 blocker psalmotoxin-1 decrease the migration and proliferation of glioma cells. PKC also abolishes the amiloride-sensitive conductance of glioma cells and inhibits hASIC1b open probability in planar lipid bilayers. In addition, hASIC1b's COOH terminus has been shown to interact with protein interacting with C kinase (PICK)1, which targets PKC to the plasma membrane. Therefore, we tested the hypothesis that PKC regulation of hASIC1b at specific PKC consensus sites inhibits hASIC1b function. We mutated three consensus PKC phosphorylation sites (T26, S40, and S499) in hASIC1b to alanine, to prevent phosphorylation, and to glutamic acid or aspartic acid, to mimic phosphorylation. Our data suggest that S40 and S499 are critical sites mediating the modulation of hASIC1b by PKC. We expressed mutant hASIC1b constructs in Xenopus oocytes and measured acid-activated currents by two-electrode voltage clamp. T26A and T26E did not exhibit acid-activated currents. S40A was indistinguishable from wild type (WT), whereas S40E, S499A, and S499D currents were decreased. The PKC activators PMA and phorbol 12,13-dibutyrate inhibited WT hASIC1b and S499A, and PMA had no effect on S40A or on WT hASIC1b in oocytes pretreated with the PKC inhibitor chelerythrine. Chelerythrine inhibited WT hASIC1b and S40A but had no effect on S499A or S40A/S499A. PKC activators or the inhibitor did not affect the surface expression of WT hASIC1b. These data show that the two PKC consensus sites S40 and S499 differentially regulate hASIC1b and mediate the effects of PKC activation or PKC inhibition on hASIC1b. This will result in a deeper understanding of PKC regulation of this channel in glioma cells, information that may help in designing potentially beneficial therapies in their treatment.
Figures

References
-
- Babinski K, Catarsi S, Biagini G, Seguela P. Mammalian ASIC2a and ASIC3 subunits co-assemble into heteromeric proton-gated channels sensitive to Gd3+. J Biol Chem 275: 28519–28525, 2000. - PubMed
-
- Baron A, Deval E, Salinas M, Lingueglia E, Voilley N, Lazdunski M. Protein kinase C stimulates the acid-sensing ion channel ASIC2a via the PDZ domain-containing protein PICK1. J Biol Chem 277: 50463–50468, 2002. - PubMed
-
- Bassilana F, Champigny G, Waldmann R, de Weille JR, Heurteaux C, Lazdunski M. The acid-sensitive ionic channel subunit ASIC and the mammalian degenerin MDEG form a heteromultimeric H+-gated Na+ channel with novel properties. J Biol Chem 272: 28819–28822, 1997. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

