WSTF regulates the H2A.X DNA damage response via a novel tyrosine kinase activity
- PMID: 19092802
- PMCID: PMC2854499
- DOI: 10.1038/nature07668
WSTF regulates the H2A.X DNA damage response via a novel tyrosine kinase activity
Abstract
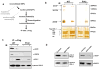
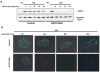
DNA double-stranded breaks present a serious challenge for eukaryotic cells. The inability to repair breaks leads to genomic instability, carcinogenesis and cell death. During the double-strand break response, mammalian chromatin undergoes reorganization demarcated by H2A.X Ser 139 phosphorylation (gamma-H2A.X). However, the regulation of gamma-H2A.X phosphorylation and its precise role in chromatin remodelling during the repair process remain unclear. Here we report a new regulatory mechanism mediated by WSTF (Williams-Beuren syndrome transcription factor, also known as BAZ1B)-a component of the WICH complex (WSTF-ISWI ATP-dependent chromatin-remodelling complex). We show that WSTF has intrinsic tyrosine kinase activity by means of a domain that shares no sequence homology to any known kinase fold. We show that WSTF phosphorylates Tyr 142 of H2A.X, and that WSTF activity has an important role in regulating several events that are critical for the DNA damage response. Our work demonstrates a new mechanism that regulates the DNA damage response and expands our knowledge of domains that contain intrinsic tyrosine kinase activity.
Figures





References
-
- Khanna KK, Jackson SP. DNA double-strand breaks: signaling, repair and the cancer connection. Nat Genet. 2001;27:247–254. - PubMed
-
- Zhou BB, Elledge SJ. The DNA damage response: putting checkpoints in perspective. Nature. 2000;408:433–439. - PubMed
-
- Redon C, et al. Histone H2A variants H2AX and H2AZ. Current opinion in genetics & development. 2002;12:162–169. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

