The changing paradigm of outflow resistance generation: towards synergistic models of the JCT and inner wall endothelium
- PMID: 19103197
- PMCID: PMC2744486
- DOI: 10.1016/j.exer.2008.11.033
The changing paradigm of outflow resistance generation: towards synergistic models of the JCT and inner wall endothelium
Abstract
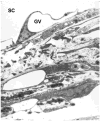
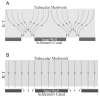
Aqueous humor outflow resistance is the primary determinant of intraocular pressure (IOP), and increased outflow resistance is the basis for elevated IOP associated with glaucoma. Experimental evidence suggests that the bulk of outflow resistance is generated in the vicinity of the inner wall endothelium of Schlemm's canal, its basement membrane and the juxtacanalicular connective tissue (JCT). However, attempts to sort out the contribution of each of these tissues to total outflow resistance have not been successful. Conventional understanding of outflow resistance assumes that the resistance of each tissue strata (i.e., the inner wall endothelium, its basement membrane and JCT) in the outflow pathway adds in series to contribute to total outflow resistance generation. However, this perspective leads to a paradox where the apparent resistances of all tissues in the outflow pathway are much lower than the measured total resistance. To resolve this paradox, we explore synergistic models of outflow resistance generation where hydrodynamic interactions between different tissue strata lead to a total resistance that is greater than the sum of the individual tissue resistances. We closely examine the "funneling" hypothesis that has emerged as a leading synergistic model, and we review the basis of funneling, mechanical and biological requirements for funneling and evidence in support of this hypothesis. We also propose refinements to the funneling model and describe how funneling may relate to segmental variability of aqueous humor outflow patterns observed within the trabecular meshwork. Pressure gradients across the JCT and inner wall endothelium will generate mechanical loads that influence the morphology of these tissues. Because tissue morphology may in turn affect outflow resistance, there exists the potential for a two-way coupling or a "fluid-solid interaction" between outflow hydrodynamics and the mechanical behavior of the inner wall and JCT. Furthermore, the adhesions and tethers between the inner wall and JCT must be physically capable of supporting such loads. We examine the structure and mechanical strength of these adhesions, and provide evidence that these adhesions and tethers are unable to support the full load imposed by the bulk of outflow resistance generation unless a substantial fraction of outflow resistance is generated within the JCT, consistent with the funneling model. This indicates that these attachments between the inner wall and JCT have considerable physiological importance for outflow resistance regulation, by maintaining the proximity between these two tissues to facilitate funneling. Further study is greatly needed to better characterize these important interactions.
Figures









References
-
- Acott TS, Kingsley PD, Samples JR, Van Buskirk EM. Human trabecular meshwork organ culture: Morphology and glycosaminoglycan synthesis. Invest Ophthalmol Vis Sci. 1988;29:90–100. - PubMed
-
- Alexopoulou AN, Multhaupt HA, Couchman JR. Syndecans in wound healing, inflammation and vascular biology. Int J Biochem Cell Biol. 2007;39:505–528. - PubMed
-
- Allingham RR, de Kater AW, Ethier CR, Anderson PJ, Hertzmark E, Epstein DL. The relationship between pore density and outflow facility in human eyes. Invest Ophthalmol Vis Sci. 1992;33:1661–1669. - PubMed
-
- Alvarado JA, Yun AJ, Murphy CG. Juxtacanalicular tissue in primary open angle glaucoma and in nonglaucomatous normals. Arch Ophthalmol. 1986;104:1517–1528. - PubMed
-
- Bahler CK, Hann CR, Fautsch MP, Johnson DH. Pharmacologic disruption of Schlemm’s canal cells and outflow facility in anterior segments of human eyes. Invest Ophthalmol Vis Sci. 2004;45:2246–2254. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

