Prevention of the pulmonary vasoconstrictor effects of HBOC-201 in awake lambs by continuously breathing nitric oxide
- PMID: 19104178
- PMCID: PMC2704389
- DOI: 10.1097/ALN.0b013e318190bc4f
Prevention of the pulmonary vasoconstrictor effects of HBOC-201 in awake lambs by continuously breathing nitric oxide
Abstract
Background: Hemoglobin-based oxygen-carrying solutions (HBOC) provide emergency alternatives to blood transfusion to carry oxygen to tissues without the risks of disease transmission or transfusion reaction. Two primary concerns hampering the clinical acceptance of acellular HBOC are the occurrence of systemic and pulmonary vasoconstriction and the maintenance of the heme-iron in the reduced state (Fe2+). We recently demonstrated that pretreatment with inhaled nitric oxide prevents the systemic hypertension induced by HBOC-201 (polymerized bovine hemoglobin) infusion in awake mice and sheep without causing methemoglobinemia. However, the impact of HBOC-201 infusion with or without inhaled nitric oxide on pulmonary vascular tone has not yet been examined.
Methods: The pulmonary and systemic hemodynamic effects of breathing nitric oxide both before and after the administration of HBOC-201 were determined in healthy, awake lambs.
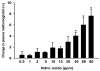
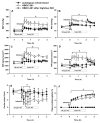
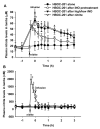
Results: Intravenous administration of HBOC-201 (12 ml/kg) induced prolonged systemic and pulmonary vasoconstriction. Pretreatment with inhaled nitric oxide (80 parts per million [ppm] for 1 h) prevented the HBOC-201--induced increase in mean arterial pressure but not the increase of pulmonary arterial pressure, systemic vascular resistance, or pulmonary vascular resistance. Pretreatment with inhaled nitric oxide (80 ppm for 1 h) followed by breathing a lower concentration of nitric oxide (5 ppm) during and after HBOC-201 infusion prevented systemic and pulmonary vasoconstriction without increasing methemoglobin levels.
Conclusions: These findings demonstrate that pretreatment with inhaled nitric oxide followed by breathing a lower concentration of the gas during and after administration of HBOC-201 may enable administration of an acellular hemoglobin substitute without vasoconstriction while preserving its oxygen-carrying capacity.
Figures


Similar articles
-
Inhaled nitric oxide enables artificial blood transfusion without hypertension.Circulation. 2008 Apr 15;117(15):1982-90. doi: 10.1161/CIRCULATIONAHA.107.729137. Epub 2008 Apr 7. Circulation. 2008. PMID: 18391111 Free PMC article.
-
Endothelial dysfunction enhances vasoconstriction due to scavenging of nitric oxide by a hemoglobin-based oxygen carrier.Anesthesiology. 2010 Mar;112(3):586-94. doi: 10.1097/ALN.0b013e3181cd7838. Anesthesiology. 2010. PMID: 20179495 Free PMC article.
-
The effects of hemoglobin glutamer-200 and iNO on pulmonary vascular tone and arterial oxygenation in an experimental acute respiratory distress syndrome.Pulm Pharmacol Ther. 2018 Apr;49:130-133. doi: 10.1016/j.pupt.2018.01.009. Epub 2018 Feb 6. Pulm Pharmacol Ther. 2018. PMID: 29425776
-
Clinical applications of inhaled nitric oxide in children with pulmonary hypertension.Adv Pharmacol. 1995;34:475-504. doi: 10.1016/s1054-3589(08)61104-7. Adv Pharmacol. 1995. PMID: 8562453 Review.
-
Systems biology of HBOC-induced vasoconstriction.Curr Drug Discov Technol. 2012 Sep;9(3):204-11. doi: 10.2174/157016312802650751. Curr Drug Discov Technol. 2012. PMID: 21726185 Free PMC article. Review.
Cited by
-
Sodium nitrite therapy attenuates the hypertensive effects of HBOC-201 via nitrite reduction.Biochem J. 2009 Aug 27;422(3):423-32. doi: 10.1042/BJ20090735. Biochem J. 2009. PMID: 19555351 Free PMC article.
-
Pulmonary hypertension in lambs transfused with stored blood is prevented by breathing nitric oxide.Anesthesiology. 2012 Mar;116(3):637-47. doi: 10.1097/ALN.0b013e318246ef77. Anesthesiology. 2012. PMID: 22293717 Free PMC article.
-
Hemoglobin infusion does not alter murine pulmonary vascular tone.Nitric Oxide. 2013 Apr 1;30:1-8. doi: 10.1016/j.niox.2012.12.007. Epub 2013 Jan 8. Nitric Oxide. 2013. PMID: 23313572 Free PMC article.
-
Development of a portable mini-generator to safely produce nitric oxide for the treatment of infants with pulmonary hypertension.Nitric Oxide. 2018 May 1;75:70-76. doi: 10.1016/j.niox.2018.02.006. Epub 2018 Feb 24. Nitric Oxide. 2018. PMID: 29486304 Free PMC article.
-
The potential adverse effects of haemolysis.Blood Transfus. 2017 May;15(3):218-221. doi: 10.2450/2017.0311-16. Blood Transfus. 2017. PMID: 28518048 Free PMC article. Review.
References
-
- Buehler PW, Alayash AI. All hemoglobin-based oxygen carriers are not created equally. Biochim Biophys Acta. 2008;1784:1378–81. - PubMed
-
- Moore EE, Johnson JL, Cheng AM, Masuno T, Banerjee A. Insights from studies of blood substitutes in trauma. Shock. 2005;24:197–205. - PubMed
-
- Reid TJ. Hb-based oxygen carriers: are we there yet? Transfusion. 2003;43:280–7. - PubMed
-
- Raat NJ, Liu JF, Doyle MP, Burhop KE, Klein J, Ince C. Effects of recombinant-hemoglobin solutions rHb2.0 and rHb1.1 on blood pressure, intestinal blood flow, and gut oxygenation in a rat model of hemorrhagic shock. J Lab Clin Med. 2005;145:21–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

