Induction of caspase- and reactive oxygen species-independent phosphatidylserine externalization in primary human neutrophils: role in macrophage recognition and engulfment
- PMID: 19106181
- PMCID: PMC2653945
- DOI: 10.1189/jlb.0408232
Induction of caspase- and reactive oxygen species-independent phosphatidylserine externalization in primary human neutrophils: role in macrophage recognition and engulfment
Abstract
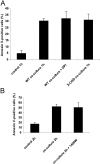
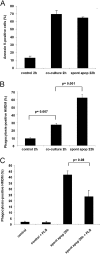
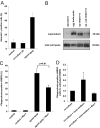
Macrophage recognition and disposal of neutrophils are important steps in the resolution of inflammation. Externalization of phosphatidylserine (PS) on the cell surface serves as a common recognition signal for macrophages and is associated with the apoptosis program in neutrophils. Here, we report that macrophage-differentiated PLB-985 cells induce rapid, caspase-independent PS externalization in human neutrophils. A similar degree of PS externalization was seen when neutrophils were cocultured with gp91(phox)-deficient PLB-985 macrophages, thus demonstrating that macrophage-induced PS externalization was NADPH oxidase-independent. Macrophage-induced PS externalization required cell-to-cell contact and kinase activation and was shown to correlate with neutrophil degranulation. Of note, the degree of engulfment of such PS-positive neutrophils by activated human monocyte-derived macrophages was considerably lower than for neutrophils undergoing constitutive apoptosis, indicating that PS externalization alone is not sufficient for macrophage disposal of neutrophils. However, addition of recombinant milk fat globule epidermal growth factor 8, a PS-binding protein, restored engulfment of the macrophage-cocultured target cells. Finally, neutrophils undergoing spontaneous apoptosis but not macrophage-cocultured neutrophils displayed surface expression and release of annexin I, and the addition of N-t-Boc-Phe-D-Leu-Phe-D-Leu-Phe (Boc1), a formyl peptide receptor/lipoxin receptor antagonist, suppressed clearance of apoptotic neutrophils. Conditioned medium from apoptotic neutrophils also promoted the engulfment of macrophage-cocultured neutrophils, and Boc1 blocked this process. Taken together, these studies highlight a novel pathway of PS externalization in primary human neutrophils and also provide evidence for an auxiliary function of annexin I in macrophage clearance of neutrophils.
Figures

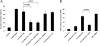

Similar articles
-
Amiloride derivatives modulate PS externalization in neutrophil-like PLB-985 cells.Biochem Pharmacol. 2010 Oct 1;80(7):1012-20. doi: 10.1016/j.bcp.2010.06.016. Epub 2010 Jun 23. Biochem Pharmacol. 2010. PMID: 20599786
-
NADPH oxidase-dependent oxidation and externalization of phosphatidylserine during apoptosis in Me2SO-differentiated HL-60 cells. Role in phagocytic clearance.J Biol Chem. 2002 Dec 20;277(51):49965-75. doi: 10.1074/jbc.M204513200. Epub 2002 Oct 9. J Biol Chem. 2002. PMID: 12376550
-
Involvement of a functional NADPH oxidase in neutrophils and macrophages during programmed cell clearance: implications for chronic granulomatous disease.Am J Physiol Cell Physiol. 2009 Sep;297(3):C621-31. doi: 10.1152/ajpcell.00651.2008. Epub 2009 Jul 1. Am J Physiol Cell Physiol. 2009. PMID: 19570889
-
Friendly fire against neutrophils: proteolytic enzymes confuse the recognition of apoptotic cells by macrophages.Biochimie. 2008 Feb;90(2):405-15. doi: 10.1016/j.biochi.2007.09.008. Epub 2007 Sep 22. Biochimie. 2008. PMID: 17964056 Review.
-
Apoptosis and macrophage clearance of neutrophils: regulation by reactive oxygen species.Redox Rep. 2003;8(3):143-50. doi: 10.1179/135100003225001511. Redox Rep. 2003. PMID: 12935311 Review.
Cited by
-
Primary phagocytosis of viable neurons by microglia activated with LPS or Aβ is dependent on calreticulin/LRP phagocytic signalling.J Neuroinflammation. 2012 Aug 13;9:196. doi: 10.1186/1742-2094-9-196. J Neuroinflammation. 2012. PMID: 22889139 Free PMC article.
-
Chelerythrine induces reactive oxygen species-dependent mitochondrial apoptotic pathway in a murine T cell lymphoma.Tumour Biol. 2014 Jan;35(1):129-40. doi: 10.1007/s13277-013-1016-4. Epub 2013 Jul 31. Tumour Biol. 2014. PMID: 23900672
-
Germline CYBB mutations that selectively affect macrophages in kindreds with X-linked predisposition to tuberculous mycobacterial disease.Nat Immunol. 2011 Mar;12(3):213-21. doi: 10.1038/ni.1992. Epub 2011 Jan 30. Nat Immunol. 2011. PMID: 21278736 Free PMC article.
-
Cell death by phagocytosis.Nat Rev Immunol. 2024 Feb;24(2):91-102. doi: 10.1038/s41577-023-00921-6. Epub 2023 Aug 21. Nat Rev Immunol. 2024. PMID: 37604896 Review.
-
IgM antibodies to oxidized phosphatidylserine as protection markers in cardiovascular disease among 60-year olds.PLoS One. 2017 Apr 21;12(4):e0171195. doi: 10.1371/journal.pone.0171195. eCollection 2017. PLoS One. 2017. PMID: 28430795 Free PMC article.
References
-
- Witasp E, Kagan V, Fadeel B. Programmed cell clearance: molecular mechanisms and role in autoimmune disease, chronic inflammation, and anti-cancer immune responses. Curr Immunol Rev. 2008;4:53–69.
-
- Lauber K, Blumenthal S G, Waibel M, Wesselborg S. Clearance of apoptotic cells: getting rid of the corpses. Mol Cell. 2004;14:277–287. - PubMed
-
- Fadok V A, Voelker D R, Campbell P A, Cohen J J, Bratton D L, Henson P M. Exposure of phosphatidylserine on the surface of apoptotic lymphocytes triggers specific recognition and removal by macrophages. J Immunol. 1992;148:2207–2216. - PubMed
-
- Fadok V A, de Cathelineau A, Daleke D L, Henson P M, Bratton D L. Loss of phospholipid asymmetry and surface exposure of phosphatidylserine is required for phagocytosis of apoptotic cells by macrophages and fibroblasts. J Biol Chem. 2001;276:1071–1077. - PubMed
-
- Kagan V E, Gleiss B, Tyurina Y Y, Tyurin V A, Elenström-Magnusson C, Liu S X, Serinkan F B, Arroyo A, Chandra J, Orrenius S, Fadeel B. A role for oxidative stress in apoptosis: oxidation and externalization of phosphatidylserine is required for macrophage clearance of cells undergoing Fas-mediated apoptosis. J Immunol. 2002;169:487–499. - PubMed

