SARS-CoV proteins decrease levels and activity of human ENaC via activation of distinct PKC isoforms
- PMID: 19112100
- PMCID: PMC2660211
- DOI: 10.1152/ajplung.90437.2008
SARS-CoV proteins decrease levels and activity of human ENaC via activation of distinct PKC isoforms
Abstract
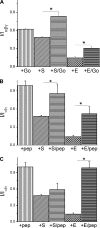
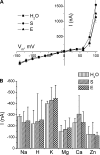
Among the multiple organ disorders caused by the severe acute respiratory syndrome coronavirus (SARS-CoV), acute lung failure following atypical pneumonia is the most serious and often fatal event. We hypothesized that two of the hydrophilic structural coronoviral proteins (S and E) would regulate alveolar fluid clearance by decreasing the cell surface expression and activity of amiloride-sensitive epithelial sodium (Na(+)) channels (ENaC), the rate-limiting protein in transepithelial Na(+) vectorial transport across distal lung epithelial cells. Coexpression of either S or E protein with human alpha-, beta-, and gamma-ENaC in Xenopus oocytes led to significant decreases of both amiloride-sensitive Na(+) currents and gamma-ENaC protein levels at their plasma membranes. S and E proteins decreased the rate of ENaC exocytosis and either had no effect (S) or decreased (E) rates of endocytosis. No direct interactions among SARS-CoV E protein with either alpha- or gamma-ENaC were indentified. Instead, the downregulation of ENaC activity by SARS proteins was partially or completely restored by administration of inhibitors of PKCalpha/beta1 and PKCzeta. Consistent with the whole cell data, expression of S and E proteins decreased ENaC single-channel activity in oocytes, and these effects were partially abrogated by PKCalpha/beta1 inhibitors. Finally, transfection of human airway epithelial (H441) cells with SARS E protein decreased whole cell amiloride-sensitive currents. These findings indicate that lung edema in SARS infection may be due at least in part to activation of PKC by SARS proteins, leading to decreasing levels and activity of ENaC at the apical surfaces of lung epithelial cells.
Figures







Similar articles
-
Regulation of epithelial sodium channel activity by SARS-CoV-1 and SARS-CoV-2 proteins.Biophys J. 2021 Jul 20;120(14):2805-2813. doi: 10.1016/j.bpj.2021.06.005. Epub 2021 Jun 29. Biophys J. 2021. PMID: 34197807 Free PMC article.
-
Characterization of a novel splice variant of δ ENaC subunit in human lungs.Am J Physiol Lung Cell Mol Physiol. 2012 Jun 15;302(12):L1262-72. doi: 10.1152/ajplung.00331.2011. Epub 2012 Apr 13. Am J Physiol Lung Cell Mol Physiol. 2012. PMID: 22505667 Free PMC article.
-
Alpha(1)-antitrypsin inhibits epithelial Na+ transport in vitro and in vivo.Am J Respir Cell Mol Biol. 2009 Sep;41(3):261-70. doi: 10.1165/rcmb.2008-0384OC. Epub 2009 Jan 8. Am J Respir Cell Mol Biol. 2009. PMID: 19131639 Free PMC article.
-
Coronavirus Proteins as Ion Channels: Current and Potential Research.Front Immunol. 2020 Oct 9;11:573339. doi: 10.3389/fimmu.2020.573339. eCollection 2020. Front Immunol. 2020. PMID: 33154751 Free PMC article. Review.
-
Expression of the severe acute respiratory syndrome coronavirus 3a protein and the assembly of coronavirus-like particles in the baculovirus expression system.Methods Mol Biol. 2007;379:35-50. doi: 10.1007/978-1-59745-393-6_3. Methods Mol Biol. 2007. PMID: 17502669 Free PMC article. Review.
Cited by
-
In Vitro and In Vivo Therapeutic Potential of 6,6'-Dihydroxythiobinupharidine (DTBN) from Nuphar lutea on Cells and K18-hACE2 Mice Infected with SARS-CoV-2.Int J Mol Sci. 2023 May 5;24(9):8327. doi: 10.3390/ijms24098327. Int J Mol Sci. 2023. PMID: 37176034 Free PMC article.
-
Protein-Protein Interactions of Viroporins in Coronaviruses and Paramyxoviruses: New Targets for Antivirals?Viruses. 2015 Jun 4;7(6):2858-83. doi: 10.3390/v7062750. Viruses. 2015. PMID: 26053927 Free PMC article. Review.
-
The Dual Role of TNF in Pulmonary Edema.J Cardiovasc Dis Res. 2010 Jan;1(1):29-36. doi: 10.4103/0975-3583.59983. J Cardiovasc Dis Res. 2010. PMID: 21188088 Free PMC article.
-
Role of epithelial sodium channels in the regulation of lung fluid homeostasis.Am J Physiol Lung Cell Mol Physiol. 2015 Dec 1;309(11):L1229-38. doi: 10.1152/ajplung.00319.2015. Epub 2015 Oct 2. Am J Physiol Lung Cell Mol Physiol. 2015. PMID: 26432872 Free PMC article. Review.
-
Is intestinal transport dysfunctional in COVID-19-related diarrhea?Am J Physiol Gastrointest Liver Physiol. 2023 May 1;324(5):G415-G418. doi: 10.1152/ajpgi.00021.2023. Epub 2023 Mar 28. Am J Physiol Gastrointest Liver Physiol. 2023. PMID: 36976797 Free PMC article.
References
-
- Burch LH, Talbot CR, Knowles MR, Canessa CM, Rossier BC, Boucher RC. Relative expression of the human epithelial Na+ channel subunits in normal and cystic fibrosis airways. Am J Physiol Cell Physiol 269: C511–C518, 1995. - PubMed
-
- Canessa CM, Schild L, Buell G, Thorens B, Gautschi I, Horisberger JD, Rossier BC. Amiloride-sensitive epithelial Na+ channel is made of three homologous subunits. Nature 367: 463–467, 1994. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

