Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes
- PMID: 19114612
- PMCID: PMC2660449
- DOI: 10.2337/dc08-1863
Sodium-glucose cotransport inhibition with dapagliflozin in type 2 diabetes
Abstract
Objective: Dapagliflozin, a novel inhibitor of renal sodium-glucose cotransporter 2, allows an insulin-independent approach to improve type 2 diabetes hyperglycemia. In this multiple-dose study we evaluated the safety and efficacy of dapagliflozin in type 2 diabetic patients.
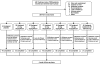
Research design and methods: Type 2 diabetic patients were randomly assigned to one of five dapagliflozin doses, metformin XR, or placebo for 12 weeks. The primary objective was to compare mean change from baseline in A1C. Other objectives included comparison of changes in fasting plasma glucose (FPG), weight, adverse events, and laboratory measurements.
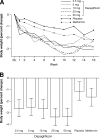
Results: After 12 weeks, dapagliflozin induced moderate glucosuria (52-85 g urinary glucose/day) and demonstrated significant glycemic improvements versus placebo (DeltaA1C -0.55 to -0.90% and DeltaFPG -16 to -31 mg/dl). Weight loss change versus placebo was -1.3 to -2.0 kg. There was no change in renal function. Serum uric acid decreased, serum magnesium increased, serum phosphate increased at higher doses, and dose-related 24-h urine volume and hematocrit increased, all of small magnitude. Treatment-emergent adverse events were similar across all groups.
Conclusions: Dapagliflozin improved hyperglycemia and facilitates weight loss in type 2 diabetic patients by inducing controlled glucosuria with urinary loss of approximately 200-300 kcal/day. Dapagliflozin treatment demonstrated no persistent, clinically significant osmolarity, volume, or renal status changes.
Trial registration: ClinicalTrials.gov NCT00263276.
Figures

References
-
- Kles KA, Vinik AI: Pathophysiology and treatment of diabetic peripheral neuropathy: the case for diabetic neurovascular function as an essential component. Curr Diabetes Rev 2: 131– 145, 2006 - PubMed
-
- Nazimek-Siewniak B, Moczulski D, Grzeszczak W: Risk of macrovascular and microvascular complications in type 2 diabetes: results of longitudinal study design. J Diabetes Complications 16: 271– 276, 2002 - PubMed
-
- Rahman S, Rahman T, Ismail AA, Rashid AR: Diabetes-associated macrovasculopathy: pathophysiology and pathogenesis. Diabetes Obes Metab 9: 767– 780, 2007 - PubMed
-
- Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HAW: 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 359: 1577– 1589, 2008 - PubMed
-
- Andrews WJ, Vasquez B, Nagulesparan M, Klimes I, Foley J, Unger R, Reaven GM: Insulin therapy in obese, non-insulin-dependent diabetes induces improvements in insulin action and secretion that are maintained for two weeks after insulin withdrawal. Diabetes 33: 634– 642, 1984 - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

