Function of Nod-like receptors in microbial recognition and host defense
- PMID: 19120480
- PMCID: PMC2679989
- DOI: 10.1111/j.1600-065X.2008.00734.x
Function of Nod-like receptors in microbial recognition and host defense
Abstract
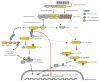
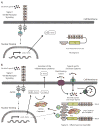
Nucleotide oligomerization domain (NOD)-like receptors (NLRs) are a specialized group of intracellular proteins that play a critical role in the regulation of the host innate immune response. NLRs act as scaffolding proteins that assemble signaling platforms that trigger nuclear factor-kappaB and mitogen-activated protein kinase signaling pathways and control the activation of inflammatory caspases. Importantly, mutations in several members of the NLR family have been linked to a variety of inflammatory diseases consistent with these molecules playing an important role in host-pathogen interactions and the inflammatory response. In this review, we focus on the role of Nod1 and Nod2 in host defense and in particular discuss recent finding regarding the role of Nlrc4, Nlpr1, and Nlrp3 inflammasomes in caspase-1 activation and subsequent release of proinflammatory cytokines such as interleukin-1 beta.
Figures

References
-
- Janeway CA, Jr, Medzhitov R. Innate immune recognition. Annu Rev Immunol. 2002;20:197–216. - PubMed
-
- Franchi L, et al. Intracellular NOD-like receptors in innate immunity, infection and disease. Cell Microbiol. 2008;10:1–8. - PubMed
-
- Sansonetti PJ. The innate signaling of dangers and the dangers of innate signaling. Nat Immunol. 2006;7:1237–1242. - PubMed
-
- Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124:783–801. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

