A multiple-method approach reveals a declining amount of chloroplast DNA during development in Arabidopsis
- PMID: 19128504
- PMCID: PMC2632658
- DOI: 10.1186/1471-2229-9-3
A multiple-method approach reveals a declining amount of chloroplast DNA during development in Arabidopsis
Abstract
Background: A decline in chloroplast DNA (cpDNA) during leaf maturity has been reported previously for eight plant species, including Arabidopsis thaliana. Recent studies, however, concluded that the amount of cpDNA during leaf development in Arabidopsis remained constant.
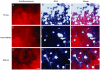
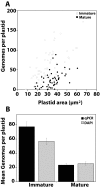
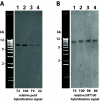
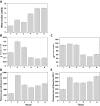
Results: To evaluate alternative hypotheses for these two contradictory observations, we examined cpDNA in Arabidopsis shoot tissues at different times during development using several methods: staining leaf sections as well as individual isolated chloroplasts with 4',6-diamidino-2-phenylindole (DAPI), real-time quantitative PCR with DNA prepared from total tissue as well as from isolated chloroplasts, fluorescence microscopy of ethidium-stained DNA molecules prepared in gel from isolated plastids, and blot-hybridization of restriction-digested total tissue DNA. We observed a developmental decline of about two- to three-fold in mean DNA per chloroplast and two- to five-fold in the fraction of cellular DNA represented by chloroplast DNA.
Conclusion: Since the two- to five-fold reduction in cpDNA content could not be attributed to an artifact of chloroplast isolation, we conclude that DNA within Arabidopsis chloroplasts is degraded in vivo as leaves mature.
Figures




References
-
- Timmis JN, Ayliffe MA, Huang CY, Martin W. Endosymbiotic gene transfer: organelle genomes forge eukaryotic chromosomes. Nat Rev Genet. 2004;5:123–135. - PubMed
-
- Kuroiwa T. The replication, differentiation, and inheritance of plastids with emphasis on the concept of organelle nuclei. Int Rev Cytol. 1991;128:1–61.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

