p53 regulates hematopoietic stem cell quiescence
- PMID: 19128791
- PMCID: PMC2839936
- DOI: 10.1016/j.stem.2008.11.006
p53 regulates hematopoietic stem cell quiescence
Abstract
The importance of the p53 protein in the cellular response to DNA damage is well known, but its function during steady-state hematopoiesis has not been established. We have defined a critical role of p53 in regulating hematopoietic stem cell quiescence, especially in promoting the enhanced quiescence seen in HSCs that lack the MEF/ELF4 transcription factor. Transcription profiling of HSCs isolated from wild-type and p53 null mice identified Gfi-1 and Necdin as p53 target genes, and using lentiviral vectors to upregulate or knockdown the expression of these genes, we show their importance in regulating HSC quiescence. Establishing the role of p53 (and its target genes) in controlling the cell-cycle entry of HSCs may lead to therapeutic strategies capable of eliminating quiescent cancer (stem) cells.
Figures



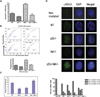
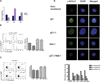
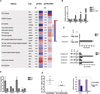
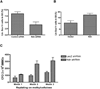
Comment in
-
Hematopoietic stem cell quiescence: yet another role for p53.Cell Stem Cell. 2009 Jan 9;4(1):7-8. doi: 10.1016/j.stem.2008.12.007. Cell Stem Cell. 2009. PMID: 19128788
References
-
- Akala OO, Park IK, Qian D, Pihalja M, Becker MW, Clarke MF. Long-term haematopoietic reconstitution by Trp53−/−p16Ink4a−/−p19Arf−/− multipotent progenitors. Nature. 2008;453:228–232. - PubMed
-
- Akashi K, Traver D, Miyamoto T, Weissman IL. A clonogenic common myeloid progenitor that gives rise to all myeloid lineages. Nature. 2000;404:193–197. - PubMed
-
- Arai F, Hirao A, Ohmura M, Sato H, Matsuoka S, Takubo K, Ito K, Koh GY, Suda T. Tie2/angiopoietin-1 signaling regulates hematopoietic stem cell quiescence in the bone marrow niche. Cell. 2004;118:149–161. - PubMed
-
- Attar EC, Scadden DT. Regulation of hematopoietic stem cell growth. Leukemia. 2004;18:1760–1768. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

