A CaV1.1 Ca2+ channel splice variant with high conductance and voltage-sensitivity alters EC coupling in developing skeletal muscle
- PMID: 19134469
- PMCID: PMC2710043
- DOI: 10.1016/j.bpj.2008.09.027
A CaV1.1 Ca2+ channel splice variant with high conductance and voltage-sensitivity alters EC coupling in developing skeletal muscle
Abstract
The Ca(2+) channel alpha(1S) subunit (Ca(V)1.1) is the voltage sensor in skeletal muscle excitation-contraction (EC) coupling. Upon membrane depolarization, this sensor rapidly triggers Ca(2+) release from internal stores and conducts a slowly activating Ca(2+) current. However, this Ca(2+) current is not essential for skeletal muscle EC coupling. Here, we identified a Ca(V)1.1 splice variant with greatly distinct current properties. The variant of the CACNA1S gene lacking exon 29 was expressed at low levels in differentiated human and mouse muscle, and up to 80% in myotubes. To test its biophysical properties, we deleted exon 29 in a green fluorescent protein (GFP)-tagged alpha(1S) subunit and expressed it in dysgenic (alpha(1S)-null) myotubes. GFP-alpha(1S)Delta 29 was correctly targeted into triads and supported skeletal muscle EC coupling. However, the Ca(2+) currents through GFP-alpha(1S)Delta 29 showed a 30-mV left-shifted voltage dependence of activation and a substantially increased open probability, giving rise to an eightfold increased current density. This robust Ca(2+) influx contributed substantially to the depolarization-induced Ca(2+) transient that triggers contraction. Moreover, deletion of exon 29 accelerated current kinetics independent of the auxiliary alpha(2)delta-1 subunit. Thus, characterizing the Ca(V)1.1 Delta 29 splice variant revealed the structural bases underlying the specific gating properties of skeletal muscle Ca(2+) channels, and it suggests the existence of a distinct mode of EC coupling in developing muscle.
Figures


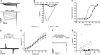

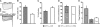
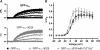
References
-
- Melzer W., Herrmann-Frank A., Luttgau H.C. The role of Ca2+ ions in excitation-contraction coupling of skeletal muscle fibres. Biochim. Biophys. Acta. 1995;1241:59–116. - PubMed
-
- Abernethy D.R., Soldatov N.M. Structure-functional diversity of human L-type Ca2+ channel: perspectives for new pharmacological targets. J. Pharmacol. Exp. Ther. 2002;300:724–728. - PubMed
-
- Perez-Reyes E., Wei X.Y., Castellano A., Birnbaumer L. Molecular diversity of L-type calcium channels. Evidence for alternative splicing of the transcripts of three non-allelic genes. J. Biol. Chem. 1990;265:20430–20436. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

