Cardiac fibroblasts require focal adhesion kinase for normal proliferation and migration
- PMID: 19136609
- PMCID: PMC2660223
- DOI: 10.1152/ajpheart.00444.2008
Cardiac fibroblasts require focal adhesion kinase for normal proliferation and migration
Abstract
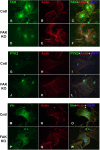
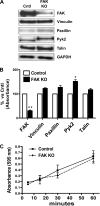
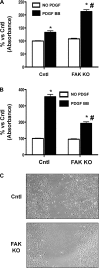
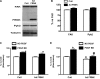
Migration and proliferation of cardiac fibroblasts (CFs) play an important role in the myocardial remodeling process. While many factors have been identified that regulate CF growth and migration, less is known about the signaling mechanisms involved in these processes. Here, we utilized Cre-LoxP technology to obtain focal adhesion kinase (FAK)-deficient adult mouse CFs and studied how FAK functioned in modulating cell adhesion, proliferation, and migration of these cells. Treatment of FAK(flox/flox) CFs with Ad/Cre virus caused over 70% reduction of FAK protein levels within a cell population. FAK-deficient CFs showed no changes in focal adhesions, cell morphology, or protein expression levels of vinculin, talin, or paxillin; proline-rich tyrosine kinase 2 (Pyk2) expression and activity were increased. Knockdown of FAK protein in CFs increased PDGF-BB-induced proliferation, while it reduced PDGF-BB-induced migration. Adhesion to fibronectin was not altered. To distinguish between the function of FAK and Pyk2, FAK function was inhibited via adenoviral-mediated overexpression of the natural FAK inhibitor FAK-related nonkinase (FRNK). Ad/FRNK had no effect on Pyk2 expression, inhibited the PDGF-BB-induced migration, but did not change the PDGF-BB-induced proliferation. FAK deficiency had only modest effects on increasing PDGF-BB activation of p38 and JNK MAPKs, with no alteration in the ERK response vs. control cells. These results demonstrate that FAK is required for the PDGF-BB-induced migratory response of adult mouse CFs and suggest that FAK could play an essential role in the wound-healing response that occurs in numerous cardiac pathologies.
Figures


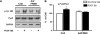
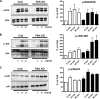
Similar articles
-
Focal adhesion kinase facilitates platelet-derived growth factor-BB-stimulated ERK2 activation required for chemotaxis migration of vascular smooth muscle cells.J Biol Chem. 2000 Dec 29;275(52):41092-9. doi: 10.1074/jbc.M005450200. J Biol Chem. 2000. PMID: 10998418
-
An endogenous inhibitor of focal adhesion kinase blocks Rac1/JNK but not Ras/ERK-dependent signaling in vascular smooth muscle cells.J Biol Chem. 2003 Aug 8;278(32):29783-91. doi: 10.1074/jbc.M303771200. Epub 2003 Jun 2. J Biol Chem. 2003. PMID: 12782622
-
Platelet-derived growth factor-BB (PDGF-BB) regulation of migration and focal adhesion kinase phosphorylation in rabbit aortic vascular smooth muscle cells: roles of phosphatidylinositol 3-kinase and mitogen-activated protein kinases.Cardiovasc Res. 1999 Mar;41(3):708-21. doi: 10.1016/s0008-6363(98)00232-6. Cardiovasc Res. 1999. PMID: 10435043
-
Tetraspanin CD9 in cell migration.Biochem Soc Trans. 2011 Apr;39(2):563-7. doi: 10.1042/BST0390563. Biochem Soc Trans. 2011. PMID: 21428940 Review.
-
Focal Adhesion's Role in Cardiomyocytes Function: From Cardiomyogenesis to Mechanotransduction.Cells. 2024 Apr 10;13(8):664. doi: 10.3390/cells13080664. Cells. 2024. PMID: 38667279 Free PMC article. Review.
Cited by
-
Integrins and integrin-related proteins in cardiac fibrosis.J Mol Cell Cardiol. 2016 Apr;93:162-74. doi: 10.1016/j.yjmcc.2015.11.010. Epub 2015 Nov 10. J Mol Cell Cardiol. 2016. PMID: 26562414 Free PMC article. Review.
-
Cardiac fibroblast: the renaissance cell.Circ Res. 2009 Dec 4;105(12):1164-76. doi: 10.1161/CIRCRESAHA.109.209809. Circ Res. 2009. PMID: 19959782 Free PMC article. Review.
-
Transcriptional control of cardiac fibroblast plasticity.J Mol Cell Cardiol. 2016 Feb;91:52-60. doi: 10.1016/j.yjmcc.2015.12.016. Epub 2015 Dec 22. J Mol Cell Cardiol. 2016. PMID: 26721596 Free PMC article. Review.
-
FAK Shutdown: Consequences on Epithelial Morphogenesis and Biomarker Expression Involving an Innovative Biomaterial for Tissue Regeneration.Int J Mol Sci. 2021 Sep 10;22(18):9774. doi: 10.3390/ijms22189774. Int J Mol Sci. 2021. PMID: 34575938 Free PMC article.
-
Vascular endothelial growth factor-C: its unrevealed role in fibrogenesis.Am J Physiol Heart Circ Physiol. 2014 Mar;306(6):H789-96. doi: 10.1152/ajpheart.00559.2013. Epub 2014 Jan 24. Am J Physiol Heart Circ Physiol. 2014. PMID: 24464750 Free PMC article.
References
-
- Banerjee I, Fuseler JW, Price RL, Borg TK, Baudino TA. Determination of cell types and numbers during cardiac development in the neonatal and adult rat and mouse. Am J Physiol Heart Circ Physiol 293: H1883–H1891, 2007. - PubMed
-
- Baudino TA, Carver W, Giles W, Borg TK. Cardiac fibroblasts: friend or foe? Am J Physiol Heart Circ Physiol 291: H1015–H1026, 2006. - PubMed
-
- Brinson AE, Harding T, Diliberto PA, He Y, Li X, Hunter D, Herman B, Earp HS, Graves LM. Regulation of a calcium-dependent tyrosine kinase in vascular smooth muscle cells by angiotensin II and platelet-derived growth factor. Dependence on calcium and the actin cytoskeleton. J Biol Chem 273: 1711–1718, 1998. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

