The SH3 domains of two PCH family members cooperate in assembly of the Schizosaccharomyces pombe contractile ring
- PMID: 19139265
- PMCID: PMC2615086
- DOI: 10.1083/jcb.200806044
The SH3 domains of two PCH family members cooperate in assembly of the Schizosaccharomyces pombe contractile ring
Abstract
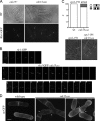
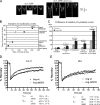
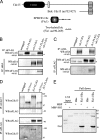
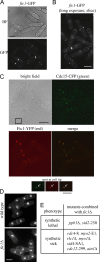
Schizosaccharomyces pombe cdc15 homology (PCH) family members participate in many cellular processes by bridging the plasma membrane and cytoskeleton. Their F-BAR domains bind and curve membranes, whereas other domains, typically SH3 domains, are expected to provide cytoskeletal links. We tested this prevailing model of functional division in the founding member of the family, Cdc15, which is essential for cytokinesis in S. pombe, and in the related PCH protein, Imp2. We find that the distinct functions of Imp2 and Cdc15 are SH3 domain independent. However, the Cdc15 and Imp2 SH3 domains share an essential role in recruiting proteins to the contractile ring, including Pxl1 and Fic1. Together, Pxl1 and Fic1, a previously uncharacterized C2 domain protein, add structural integrity to the contractile ring and prevent it from fragmenting during division. Our data indicate that the F-BAR proteins Cdc15 and Imp2 contribute to a single biological process with both distinct and overlapping functions.
Figures





Similar articles
-
The Cdc15 and Imp2 SH3 domains cooperatively scaffold a network of proteins that redundantly ensure efficient cell division in fission yeast.Mol Biol Cell. 2015 Jan 15;26(2):256-69. doi: 10.1091/mbc.E14-10-1451. Epub 2014 Nov 26. Mol Biol Cell. 2015. PMID: 25428987 Free PMC article.
-
The intrinsically disordered region of the cytokinetic F-BAR protein Cdc15 performs a unique essential function in maintenance of cytokinetic ring integrity.Mol Biol Cell. 2019 Oct 15;30(22):2790-2801. doi: 10.1091/mbc.E19-06-0314. Epub 2019 Sep 11. Mol Biol Cell. 2019. PMID: 31509478 Free PMC article.
-
Cooperation between Paxillin-like Protein Pxl1 and Glucan Synthase Bgs1 Is Essential for Actomyosin Ring Stability and Septum Formation in Fission Yeast.PLoS Genet. 2015 Jul 1;11(7):e1005358. doi: 10.1371/journal.pgen.1005358. eCollection 2015 Jul. PLoS Genet. 2015. PMID: 26132084 Free PMC article.
-
Pombe Cdc15 homology proteins: regulators of membrane dynamics and the actin cytoskeleton.Trends Biochem Sci. 2006 Dec;31(12):670-9. doi: 10.1016/j.tibs.2006.10.001. Epub 2006 Oct 30. Trends Biochem Sci. 2006. PMID: 17074490 Review.
-
Roles of F-BAR/PCH proteins in the regulation of membrane dynamics and actin reorganization.Int Rev Cell Mol Biol. 2009;272:1-31. doi: 10.1016/S1937-6448(08)01601-8. Int Rev Cell Mol Biol. 2009. PMID: 19121815 Review.
Cited by
-
Predicting the fission yeast protein interaction network.G3 (Bethesda). 2012 Apr;2(4):453-67. doi: 10.1534/g3.111.001560. Epub 2012 Apr 1. G3 (Bethesda). 2012. PMID: 22540037 Free PMC article.
-
SH3Ps-Evolution and Diversity of a Family of Proteins Engaged in Plant Cytokinesis.Int J Mol Sci. 2019 Nov 11;20(22):5623. doi: 10.3390/ijms20225623. Int J Mol Sci. 2019. PMID: 31717902 Free PMC article.
-
Contractile ring stability in S. pombe depends on F-BAR protein Cdc15p and Bgs1p transport from the Golgi complex.Cell Rep. 2014 Sep 11;8(5):1533-44. doi: 10.1016/j.celrep.2014.07.048. Epub 2014 Aug 21. Cell Rep. 2014. PMID: 25159149 Free PMC article.
-
Hof1 and Rvs167 have redundant roles in actomyosin ring function during cytokinesis in budding yeast.PLoS One. 2013;8(2):e57846. doi: 10.1371/journal.pone.0057846. Epub 2013 Feb 28. PLoS One. 2013. PMID: 23469085 Free PMC article.
-
Multiple polarity kinases inhibit phase separation of F-BAR protein Cdc15 and antagonize cytokinetic ring assembly in fission yeast.Elife. 2023 Feb 7;12:e83062. doi: 10.7554/eLife.83062. Elife. 2023. PMID: 36749320 Free PMC article.
References
-
- Arai, R., and I. Mabuchi. 2002. F-actin ring formation and the role of F-actin cables in the fission yeast Schizosaccharomyces pombe. J. Cell Sci. 115:887–898. - PubMed
-
- Bahler, J., J.Q. Wu, M.S. Longtine, N.G. Shah, A. McKenzie III, A.B. Steever, A. Wach, P. Philippsen, and J.R. Pringle. 1998. Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast. 14:943–951. - PubMed
-
- Barr, F.A., and U. Gruneberg. 2007. Cytokinesis: placing and making the final cut. Cell. 131:847–860. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

