Aging-related loss of the chromatin protein HMGB2 in articular cartilage is linked to reduced cellularity and osteoarthritis
- PMID: 19139395
- PMCID: PMC2633567
- DOI: 10.1073/pnas.0806062106
Aging-related loss of the chromatin protein HMGB2 in articular cartilage is linked to reduced cellularity and osteoarthritis
Abstract
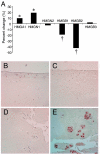
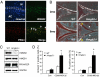
Osteoarthritis (OA) is the most common joint disease and typically begins with an aging-related disruption of the articular cartilage surface. Mechanisms leading to the aging-related cartilage surface degeneration remain to be determined. Here, we demonstrate that nonhistone chromatin protein high-mobility group box (HMGB) protein 2 is uniquely expressed in the superficial zone (SZ) of human articular cartilage. In human and murine cartilage, there is an aging-related loss of HMGB2 expression, ultimately leading to its complete absence. Mice genetically deficient in HMGB2 (Hmgb2(-/-)) show earlier onset of and more severe OA. This is associated with a profound reduction in cartilage cellularity attributable to increased cell death. These cellular changes precede glycosaminoglycan depletion and progressive cartilage erosions. Chondrocytes from Hmgb2(-/-) mice are more susceptible to apoptosis induction in vitro. In conclusion, HMGB2 is a transcriptional regulator specifically expressed in the SZ of human articular cartilage and supports chondrocyte survival. Aging is associated with a loss of HMGB2 expression and reduced cellularity, and this contributes to the development of OA.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Felson DT, et al. The prevalence of knee osteoarthritis in the elderly. The Framingham Osteoarthritis Study. Arthritis Rheum. 1987;30:914–918. - PubMed
-
- Sharma L, Kapoor D. In: Osteoarthritis. Moskowitz R, editor. Philadelphia: Wolters Kluwer; 2007.
-
- Bierma-Zeinstra SM, Koes BW. Risk factors and prognostic factors of hip and knee osteoarthritis. Nature Clinical Practice Rheumatology. 2007;3:78–85. - PubMed
-
- Poole AR, Guilak F, Abramson S. In: Osteoarthritis. Moskowitz R, editor. Philadelphia: Wolters Kluwer; 2007.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

