Striatal readministration of rAAV vectors reveals an immune response against AAV2 capsids that can be circumvented
- PMID: 19142181
- PMCID: PMC2835093
- DOI: 10.1038/mt.2008.284
Striatal readministration of rAAV vectors reveals an immune response against AAV2 capsids that can be circumvented
Abstract
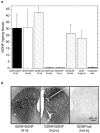
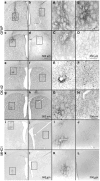
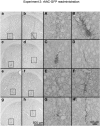
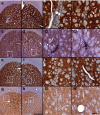
Recombinant adeno-associated virus (rAAV) expresses no viral genes after transduction. In addition, because the brain is relatively immunoprivileged, intracranial rAAV transduction may be immunologically benign due to a lack of antigen presentation. However, preexposure to AAV allows neutralizing antibodies (nAbs) to block brain transduction and rAAV readministration in the brain leads to an inflammatory response in the second-injection site. In this study, we replicate our striatal rAAV2/2-GDNF readministration results and extend this effect to a second transgene, green fluorescent protein (GFP). Unlike rAAV2/2-GDNF readministration, striatal rAAV2/2-GFP readministration leads to a loss of transgene in the second site in the absence of detectable circulating nAbs. In order to determine whether the transgene or the AAV2 capsid is the antigenic stimulus in brain for the immune response in the second site, we readministered rAAV2/2-GFP using two different rAAV serotypes (rAAV2/2 followed by rAAV2/5). In this case, there was no striatal inflammation or transgene loss detected in the second-injection site. In addition, striatal readministration of rAAV2/5-GFP also resulted in no detectable immune response. Furthermore, delaying rAAV2/2 striatal readministration to a 11-week interval abrogated the immune response in the second-injection site. Finally, while striatal readministration of rAAV2/2 leads to significant loss of transgene in the second-injection site, this effect is not due to loss of vector genomes as determined by quantitative real-time PCR. We conclude that intracellular processing of AAV capsids after transduction is the immunogenic antigen and capsid serotypes that are processed more quickly than rAAV2/2 are less immunogenic.
Figures




Similar articles
-
Circulating anti-wild-type adeno-associated virus type 2 (AAV2) antibodies inhibit recombinant AAV2 (rAAV2)-mediated, but not rAAV5-mediated, gene transfer in the brain.J Virol. 2004 Jun;78(12):6344-59. doi: 10.1128/JVI.78.12.6344-6359.2004. J Virol. 2004. PMID: 15163728 Free PMC article.
-
Time course of transgene expression after intrastriatal pseudotyped rAAV2/1, rAAV2/2, rAAV2/5, and rAAV2/8 transduction in the rat.Mol Ther. 2007 Aug;15(8):1504-11. doi: 10.1038/sj.mt.6300227. Epub 2007 Jun 12. Mol Ther. 2007. PMID: 17565350
-
Transient immunosuppression allows transgene expression following readministration of adeno-associated viral vectors.Hum Gene Ther. 1998 Mar 1;9(4):477-85. doi: 10.1089/hum.1998.9.4-477. Hum Gene Ther. 1998. PMID: 9525309
-
Recombinant AAV-mediated gene delivery to the central nervous system.J Gene Med. 2004 Feb;6 Suppl 1:S212-22. doi: 10.1002/jgm.506. J Gene Med. 2004. PMID: 14978764 Review.
-
Tissue and cell-type-specific transduction using rAAV vectors in lung diseases.J Mol Med (Berl). 2021 Aug;99(8):1057-1071. doi: 10.1007/s00109-021-02086-y. Epub 2021 May 21. J Mol Med (Berl). 2021. PMID: 34021360 Review.
Cited by
-
Adeno-associated viral vector serotype 5 poorly transduces liver in rat models.PLoS One. 2013 Dec 27;8(12):e82597. doi: 10.1371/journal.pone.0082597. eCollection 2013. PLoS One. 2013. PMID: 24386104 Free PMC article.
-
Pre-immunization with an intramuscular injection of AAV9-human erythropoietin vectors reduces the vector-mediated transduction following re-administration in rat brain.PLoS One. 2013 May 8;8(5):e63876. doi: 10.1371/journal.pone.0063876. Print 2013. PLoS One. 2013. PMID: 23667683 Free PMC article.
-
The Long and Winding Road: From the High-Affinity Choline Uptake Site to Clinical Trials for Malignant Brain Tumors.Adv Pharmacol. 2016;76:147-73. doi: 10.1016/bs.apha.2016.03.002. Epub 2016 Apr 23. Adv Pharmacol. 2016. PMID: 27288077 Free PMC article. Review.
-
Adeno-Associated Virus Neutralizing Antibodies in Large Animals and Their Impact on Brain Intraparenchymal Gene Transfer.Mol Ther Methods Clin Dev. 2018 Oct 4;11:65-72. doi: 10.1016/j.omtm.2018.09.003. eCollection 2018 Dec 14. Mol Ther Methods Clin Dev. 2018. PMID: 30397628 Free PMC article.
-
Laminin-coated electronic scaffolds with vascular topography for tracking and promoting the migration of brain cells after injury.Nat Biomed Eng. 2023 Oct;7(10):1282-1292. doi: 10.1038/s41551-023-01101-6. Epub 2023 Oct 9. Nat Biomed Eng. 2023. PMID: 37814007
References
-
- Lowenstein PR, Mandel RJ, Xiong WD, Kroeger K., and , Castro MG. Immune responses to adenovirus and adeno-associated vectors used for gene therapy of brain diseases: the role of immunological synapses in understanding the cell biology of neuroimmune interactions. Curr Gene Ther. 2007;7:347–360. - PMC - PubMed
-
- Mandel RJ, Manfredsson FP, Foust KD, Rising A, Reimsnider S, Nash K, et al. Recombinant adeno-associated viral vectors as therapeutic agents to treat neurological disorders. Mol Ther. 2006;13:463–483. - PubMed
-
- Daadi MM, Pivirotto P, Bringas J, Cunningham J, Forsayeth J, Eberling J, et al. Distribution of AAV2-hAADC-transduced cells after 3 years in Parkinsonian monkeys. Neuroreport. 2006;17:201–204. - PubMed
-
- Bankiewicz KS, Forsayeth J, Eberling JL, Sanchez-Pernaute R, Pivirotto P, Bringas J, et al. Long-term clinical improvement in MPTP-lesioned primates after gene therapy with AAV-hAADC. Mol Ther. 2006;14:564–570. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

