Lipopolysaccharide-induced osteoclastogenesis from mononuclear precursors: a mechanism for osteolysis in chronic otitis
- PMID: 19145462
- PMCID: PMC2674198
- DOI: 10.1007/s10162-008-0153-8
Lipopolysaccharide-induced osteoclastogenesis from mononuclear precursors: a mechanism for osteolysis in chronic otitis
Abstract
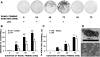
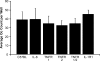
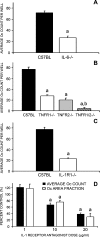
Osteoclasts are the only cells capable of carrying out bone resorption and therefore are responsible for the osteolysis seen in infectious diseases such as chronic otitis media and infected cholesteatoma. Pseudomonas aeruginosa is the most common organism isolated from these infectious middle ear diseases. In this study, we examined the mechanisms by which P. aeruginosa lipopolysaccharide (LPS) stimulates osteoclastogenesis directly from mononuclear osteoclast precursor cells. Osteoclast precursors demonstrated robust, bone-resorbing osteoclast formation when stimulated by P. aeruginosa LPS only if previously primed with permissive, sub-osteoclastogenic doses of receptor activator of NF-kappaB ligand (RANKL), suggesting that LPS is osteoclastogenic only during a specific developmental window. Numerous LPS-elicited cytokines were found to be released by osteoclast precursors undergoing P. aeruginosa LPS-mediated osteoclast formation. Two lines of evidence suggest that several cytokines promote Oc formation in an autocrine/paracrine manner. First, inhibition of several cytokine pathways including TNF-alpha, IL-1, and IL-6 block the osteoclastogenesis induced by LPS. Secondly, increased expression of the receptors for TNF-alpha and IL-1 was demonstrated by real-time quantitative polymerase chain reaction. Such a mechanism has not previously been established and demonstrates the ability of osteoclast precursors to autonomously facilitate bone destruction.
Figures


Similar articles
-
Sesquiterpene lactone parthenolide blocks lipopolysaccharide-induced osteolysis through the suppression of NF-kappaB activity.J Bone Miner Res. 2004 Nov;19(11):1905-16. doi: 10.1359/JBMR.040919. Epub 2004 Sep 27. J Bone Miner Res. 2004. PMID: 15476591
-
Dual modulation of osteoclast differentiation by lipopolysaccharide.J Bone Miner Res. 2002 Jul;17(7):1211-8. doi: 10.1359/jbmr.2002.17.7.1211. J Bone Miner Res. 2002. PMID: 12096834
-
The protective effect of WKYMVm peptide on inflammatory osteolysis through regulating NF-κB and CD9/gp130/STAT3 signalling pathway.J Cell Mol Med. 2020 Jan;24(2):1893-1905. doi: 10.1111/jcmm.14885. Epub 2019 Dec 14. J Cell Mol Med. 2020. PMID: 31837208 Free PMC article.
-
Diallyl disulfide alleviates inflammatory osteolysis by suppressing osteoclastogenesis via NF-κB-NFATc1 signal pathway.FASEB J. 2019 Jun;33(6):7261-7273. doi: 10.1096/fj.201802172R. Epub 2019 Mar 11. FASEB J. 2019. PMID: 30857415 Free PMC article.
-
[RANKL signaling and bone diseases. Quiescent osteoclast precursors and RANKL signaling].Clin Calcium. 2011 Aug;21(8):1187-92. Clin Calcium. 2011. PMID: 21814024 Review. Japanese.
Cited by
-
Osteoclastogenesis in periodontal diseases: Possible mediators and mechanisms.J Oral Biosci. 2020 Jun;62(2):123-130. doi: 10.1016/j.job.2020.02.002. Epub 2020 Feb 17. J Oral Biosci. 2020. PMID: 32081710 Free PMC article. Review.
-
METTL3 Regulates Osteoclast Biological Behaviors via iNOS/NO-Mediated Mitochondrial Dysfunction in Inflammatory Conditions.Int J Mol Sci. 2023 Jan 11;24(2):1403. doi: 10.3390/ijms24021403. Int J Mol Sci. 2023. PMID: 36674918 Free PMC article.
-
The Neuroimmune Interplay in Joint Pain: The Role of Macrophages.Front Immunol. 2022 Mar 10;13:812962. doi: 10.3389/fimmu.2022.812962. eCollection 2022. Front Immunol. 2022. PMID: 35355986 Free PMC article. Review.
-
The role of bone resorption in the etiopathogenesis of acquired middle ear cholesteatoma.Eur Arch Otorhinolaryngol. 2017 May;274(5):2071-2078. doi: 10.1007/s00405-016-4422-6. Epub 2016 Dec 16. Eur Arch Otorhinolaryngol. 2017. PMID: 27981350 Review.
-
TLR4 drives the pathogenesis of acquired cholesteatoma by promoting local inflammation and bone destruction.Sci Rep. 2015 Dec 7;5:16683. doi: 10.1038/srep16683. Sci Rep. 2015. PMID: 26639190 Free PMC article.
References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1172/JCI119679', 'is_inner': False, 'url': 'https://doi.org/10.1172/jci119679'}, {'type': 'PMC', 'value': 'PMC508337', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC508337/'}, {'type': 'PubMed', 'value': '9294124', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/9294124/'}]}
- Abu-Amer Y, Ross FP, et al. Lipopolysaccharide-stimulated osteoclastogenesis is mediated by tumor necrosis factor via its P55 receptor. J. Clin. Invest. 100(6):1557–1565, 1997. - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1038/416686a', 'is_inner': False, 'url': 'https://doi.org/10.1038/416686a'}, {'type': 'PubMed', 'value': '11961535', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/11961535/'}]}
- Alliston T, Derynck R. Medicine: interfering with bone remodelling. Nature 416(6882):686–687, 2002. - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1074/jbc.275.7.4858', 'is_inner': False, 'url': 'https://doi.org/10.1074/jbc.275.7.4858'}, {'type': 'PubMed', 'value': '10671521', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/10671521/'}]}
- Azuma Y, Kaji K, et al. Tumor necrosis factor-alpha induces differentiation of and bone resorption by osteoclasts. J. Biol. Chem. 275(7):4858–4864, 2000. - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1677/joe.0.1750405', 'is_inner': False, 'url': 'https://doi.org/10.1677/joe.0.1750405'}, {'type': 'PubMed', 'value': '12429038', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/12429038/'}]}
- Cornish J, Callon KE, et al. Leptin directly regulates bone cell function in vitro and reduces bone fragility in vivo. J. Endocrinol. 175(2):405–415, 2002. - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1038/372560a0', 'is_inner': False, 'url': 'https://doi.org/10.1038/372560a0'}, {'type': 'PubMed', 'value': '7990930', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/7990930/'}]}
- Erickson SL, de Sauvage FJ, et al. Decreased sensitivity to tumour-necrosis factor but normal T-cell development in TNF receptor-2-deficient mice. Nature 372(6506):560–563, 1994. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

