Role of basic leucine zipper proteins in transcriptional regulation of the steroidogenic acute regulatory protein gene
- PMID: 19150388
- PMCID: PMC5006949
- DOI: 10.1016/j.mce.2008.12.009
Role of basic leucine zipper proteins in transcriptional regulation of the steroidogenic acute regulatory protein gene
Abstract
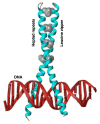
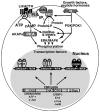
The regulation of steroidogenic acute regulatory protein (StAR) gene transcription by cAMP-dependent mechanisms occurs in the absence of a consensus cAMP response element (CRE, TGACGTGA). This regulation is coordinated by multiple transcription factors that bind to sequence-specific elements located approximately 150 bp upstream of the transcription start site. Among the proteins that bind within this region, the basic leucine zipper (bZIP) family of transcription factors, i.e. CRE binding protein (CREB)/CRE modulator (CREM)/activating transcription factor (ATF), activator protein 1 (AP-1; Fos/Jun), and CCAAT enhancer binding protein beta (C/EBPbeta), interact with an overlapping region (-81/-72 bp) in the StAR promoter, mediate stimulus-transcription coupling of cAMP signaling and play integral roles in regulating StAR gene expression. These bZIP proteins are structurally similar and bind to DNA sequences as dimers; however, they exhibit discrete transcriptional activities, interact with several transcription factors and other properties that contribute in their regulatory functions. The 5'-flanking -81/-72 bp region of the StAR gene appears to function as a key element within a complex cAMP response unit by binding to different bZIP members, and the StAR promoter displays variable states of cAMP responsivity contingent upon the occupancy of these cis-elements with these transcription factors. The expression and activities of CREB/CREM/ATF, Fos/Jun and C/EBPbeta have been demonstrated to be mediated by a plethora of extracellular signals, and the phosphorylation of these proteins at several Ser and Thr residues allows recruitment of the transcriptional coactivator CREB binding protein (CBP) or its functional homolog p300 to the StAR promoter. This review will focus on the current level of understanding of the roles of selective bZIP family proteins within the complex series of processes involved in regulating StAR gene transcription.
Figures


Similar articles
-
Crosstalk of CREB and Fos/Jun on a single cis-element: transcriptional repression of the steroidogenic acute regulatory protein gene.J Mol Endocrinol. 2007 Oct;39(4):261-77. doi: 10.1677/JME-07-0065. J Mol Endocrinol. 2007. PMID: 17909266
-
Regulation of steroidogenesis and the steroidogenic acute regulatory protein by a member of the cAMP response-element binding protein family.Mol Endocrinol. 2002 Jan;16(1):184-99. doi: 10.1210/mend.16.1.0759. Mol Endocrinol. 2002. PMID: 11773448
-
Assessment of the role of activator protein-1 on transcription of the mouse steroidogenic acute regulatory protein gene.Mol Endocrinol. 2004 Mar;18(3):558-73. doi: 10.1210/me.2003-0223. Epub 2003 Dec 12. Mol Endocrinol. 2004. PMID: 14673133
-
The molecular biology and nomenclature of the activating transcription factor/cAMP responsive element binding family of transcription factors: activating transcription factor proteins and homeostasis.Gene. 2001 Jul 25;273(1):1-11. doi: 10.1016/s0378-1119(01)00551-0. Gene. 2001. PMID: 11483355 Review.
-
Involvement of multiple transcription factors in the regulation of steroidogenic acute regulatory protein gene expression.Steroids. 2003 Dec;68(14):1125-34. doi: 10.1016/j.steroids.2003.07.009. Steroids. 2003. PMID: 14643873 Review.
Cited by
-
A fish with no sex: gonadal and adrenal functions partition between zebrafish NR5A1 co-orthologs.Genetics. 2021 Feb 9;217(2):iyaa030. doi: 10.1093/genetics/iyaa030. Genetics. 2021. PMID: 33724412 Free PMC article.
-
Expression and Function of StAR in Cancerous and Non-Cancerous Human and Mouse Breast Tissues: New Insights into Diagnosis and Treatment of Hormone-Sensitive Breast Cancer.Int J Mol Sci. 2023 Jan 1;24(1):758. doi: 10.3390/ijms24010758. Int J Mol Sci. 2023. PMID: 36614200 Free PMC article.
-
Modeling bidirectional transcription using silkmoth chorion gene promoters.Organogenesis. 2010 Jan-Mar;6(1):54-8. doi: 10.4161/org.6.1.10696. Organogenesis. 2010. PMID: 20592866 Free PMC article. Review.
-
The involvement of specific PKC isoenzymes in phorbol ester-mediated regulation of steroidogenic acute regulatory protein expression and steroid synthesis in mouse Leydig cells.Endocrinology. 2011 Jan;152(1):313-25. doi: 10.1210/en.2010-0874. Epub 2010 Nov 3. Endocrinology. 2011. PMID: 21047949 Free PMC article.
-
A narrative review on inflammaging and late-onset hypogonadism.Front Endocrinol (Lausanne). 2024 Jan 17;15:1291389. doi: 10.3389/fendo.2024.1291389. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 38298378 Free PMC article. Review.
References
-
- Angel P, Karin M. The role of Jun, Fos and the AP-1 complex in cell-proliferation and transformation. Biochim Biophys Acta. 1991;1072:129–157. - PubMed
-
- Arias J, Alberts AS, Brindle P, Claret FX, Smeal T, Karin M, Feramisco J, Montminy M. Activation of cAMP and mitogen responsive genes relies on a common nuclear factor. Nature. 1994;370:226–229. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

