Signaling to the apical membrane and to the paracellular pathway: changes in the cytosolic proteome of Aedes Malpighian tubules
- PMID: 19151207
- PMCID: PMC2727076
- DOI: 10.1242/jeb.024646
Signaling to the apical membrane and to the paracellular pathway: changes in the cytosolic proteome of Aedes Malpighian tubules
Abstract
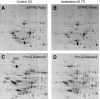
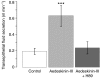
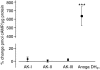
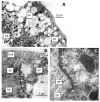
Using a proteomics approach, we examined the post-translational changes in cytosolic proteins when isolated Malpighian tubules of Aedes aegypti were stimulated for 1 min with the diuretic peptide aedeskinin-III (AK-III, 10(-7) mol l(-1)). The cytosols of control (C) and aedeskinin-treated (T) tubules were extracted from several thousand Malpighian tubules, subjected to 2-D electrophoresis and stained for total proteins and phosphoproteins. The comparison of C and T gels was performed by gel image analysis for the change of normalized spot volumes. Spots with volumes equal to or exceeding C/T ratios of +/-1.5 were robotically picked for in-gel digestion with trypsin and submitted for protein identification by nanoLC/MS/MS analysis. Identified proteins covered a wide range of biological activity. As kinin peptides are known to rapidly stimulate transepithelial secretion of electrolytes and water by Malpighian tubules, we focused on those proteins that might mediate the increase in transepithelial secretion. We found that AK-III reduces the cytosolic presence of subunits A and B of the V-type H(+) ATPase, endoplasmin, calreticulin, annexin, type II regulatory subunit of protein kinase A (PKA) and rab GDP dissociation inhibitor and increases the cytosolic presence of adducin, actin, Ca(2+)-binding protein regucalcin/SMP30 and actin-depolymerizing factor. Supporting the putative role of PKA in the AK-III-induced activation of the V-type H(+) ATPase is the effect of H89, an inhibitor of PKA, on fluid secretion. H89 reverses the stimulatory effect of AK-III on transepithelial fluid secretion in isolated Malpighian tubules. However, AK-III does not raise intracellular levels of cAMP, the usual activator of PKA, suggesting a cAMP-independent activation of PKA that removes subunits A and B from the cytoplasm in the assembly and activation of the V-type H(+) ATPase. Alternatively, protein kinase C could also mediate the activation of the proton pump. Ca(2+) remains the primary intracellular messenger of the aedeskinins that signals the remodeling of the paracellular complex apparently through protein kinase C, thereby increasing transepithelial anion secretion. The effects of AK-III on active transcellular and passive paracellular transport are additive, if not synergistic, to bring about the rapid diuresis.
Figures


Similar articles
-
Transcellular and paracellular pathways of transepithelial fluid secretion in Malpighian (renal) tubules of the yellow fever mosquito Aedes aegypti.Acta Physiol (Oxf). 2011 Jul;202(3):387-407. doi: 10.1111/j.1748-1716.2010.02195.x. Epub 2010 Nov 16. Acta Physiol (Oxf). 2011. PMID: 20946239 Free PMC article. Review.
-
A SLC4-like anion exchanger from renal tubules of the mosquito (Aedes aegypti): evidence for a novel role of stellate cells in diuretic fluid secretion.Am J Physiol Regul Integr Comp Physiol. 2010 Mar;298(3):R642-60. doi: 10.1152/ajpregu.00729.2009. Epub 2009 Dec 30. Am J Physiol Regul Integr Comp Physiol. 2010. PMID: 20042685 Free PMC article.
-
The single kinin receptor signals to separate and independent physiological pathways in Malpighian tubules of the yellow fever mosquito.Am J Physiol Regul Integr Comp Physiol. 2010 Aug;299(2):R612-22. doi: 10.1152/ajpregu.00068.2010. Epub 2010 Jun 10. Am J Physiol Regul Integr Comp Physiol. 2010. PMID: 20538895 Free PMC article.
-
Roles of PKC and phospho-adducin in transepithelial fluid secretion by Malpighian tubules of the yellow fever mosquito.Tissue Barriers. 2013 Jan 1;1(1):e23120. doi: 10.4161/tisb.23120. Tissue Barriers. 2013. PMID: 24062972 Free PMC article.
-
Transport mechanisms of diuresis in Malpighian tubules of insects.J Exp Biol. 2003 Nov;206(Pt 21):3845-56. doi: 10.1242/jeb.00639. J Exp Biol. 2003. PMID: 14506220 Review.
Cited by
-
Transcellular and paracellular pathways of transepithelial fluid secretion in Malpighian (renal) tubules of the yellow fever mosquito Aedes aegypti.Acta Physiol (Oxf). 2011 Jul;202(3):387-407. doi: 10.1111/j.1748-1716.2010.02195.x. Epub 2010 Nov 16. Acta Physiol (Oxf). 2011. PMID: 20946239 Free PMC article. Review.
-
A dynamic paracellular pathway serves diuresis in mosquito Malpighian tubules.Ann N Y Acad Sci. 2012 Jul;1258(1):166-76. doi: 10.1111/j.1749-6632.2012.06527.x. Ann N Y Acad Sci. 2012. PMID: 22731730 Free PMC article.
-
Cloning and functional characterization of inward-rectifying potassium (Kir) channels from Malpighian tubules of the mosquito Aedes aegypti.Insect Biochem Mol Biol. 2013 Jan;43(1):75-90. doi: 10.1016/j.ibmb.2012.09.009. Epub 2012 Oct 17. Insect Biochem Mol Biol. 2013. PMID: 23085358 Free PMC article.
-
A SLC4-like anion exchanger from renal tubules of the mosquito (Aedes aegypti): evidence for a novel role of stellate cells in diuretic fluid secretion.Am J Physiol Regul Integr Comp Physiol. 2010 Mar;298(3):R642-60. doi: 10.1152/ajpregu.00729.2009. Epub 2009 Dec 30. Am J Physiol Regul Integr Comp Physiol. 2010. PMID: 20042685 Free PMC article.
-
Chloride channels in stellate cells are essential for uniquely high secretion rates in neuropeptide-stimulated Drosophila diuresis.Proc Natl Acad Sci U S A. 2014 Sep 30;111(39):14301-6. doi: 10.1073/pnas.1412706111. Epub 2014 Sep 16. Proc Natl Acad Sci U S A. 2014. PMID: 25228763 Free PMC article.
References
-
- Anderson, J. M. and Van Itallie, C. M. (1995). Tight junctions and the molecular basis for regulation of paracellular permeability. Am. J. Physiol. 269, G467-G475. - PubMed
-
- Behr, M., Riedel, D. and Schuh, R. (2003). The claudin-like megatrachea is essential in septate junctions for the epithelial barrier function in Drosophila. Dev. Cell 5, 611-620. - PubMed
-
- Beltran, C. and Nelson, N. (1992). The membrane sector of vauolar H+-ATPase by itself is impermeable to protons. Acta Physiol. Scand. Suppl. 607, 41-47. - PubMed
-
- Belvin, M. P., Jin, Y. and Anderson, K. V. (1995). Cactus protein degradation mediates Drosophila dorsal-ventral signaling. Genes Dev. 9, 783-793. - PubMed
-
- Beyenbach, K. W. (2001). Energizing epithelial transport with the vacuolar H+-ATPase. News Physiol. Sci. 16, 145-151. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

