Critical role of serum response factor in pulmonary myofibroblast differentiation induced by TGF-beta
- PMID: 19151320
- PMCID: PMC2742753
- DOI: 10.1165/rcmb.2008-0288OC
Critical role of serum response factor in pulmonary myofibroblast differentiation induced by TGF-beta
Abstract
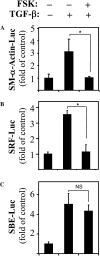
Transforming growth factor-beta (TGF-beta) is a cytokine implicated in wound healing and in the pathogenesis of pulmonary fibrosis. TGF-beta stimulates myofibroblast differentiation characterized by expression of contractile smooth muscle (SM)-specific proteins such as SM-alpha-actin. In the present study, we examined the role of serum response factor (SRF) in the mechanism of TGF-beta-induced pulmonary myofibroblast differentiation of human lung fibroblasts (HLF). TGF-beta stimulated SM-alpha-actin expression in HLF, which paralleled with a profound induction of SRF expression and activity. Inhibition of SRF by the pharmacologic SRF inhibitor (CCG-1423), or via adenovirus-mediated transduction of SRF short hairpin RNA (shSRF), blocked the expression of both SRF and SM-alpha-actin in response to TGF-beta without affecting Smad-mediated signaling of TGF-beta. However, forced expression of SRF on its own did not promote SM-alpha-actin expression, whereas expression of the constitutively transactivated SRF fusion protein (SRF-VP16) was sufficient to induce SM-alpha-actin expression, suggesting that both expression and transactivation of SRF are important. Activation of protein kinase A (PKA) by forskolin or iloprost resulted in a significant inhibition of SM-alpha-actin expression induced by TGF-beta, and this was associated with inhibition of both SRF expression and activity, but not of Smad-mediated gene transcription. In summary, this is the first direct demonstration that TGF-beta-induced pulmonary myofibroblast differentiation is mediated by SRF, and that inhibition of myofibroblast differentiation by PKA occurs through down-regulation of SRF expression levels and SRF activity, independent of Smad signaling.
Figures





References
-
- Leask A, Abraham DJ. TGF-beta signaling and the fibrotic response. FASEB J 2004;18:816–827. - PubMed
-
- Phan SH. Fibroblast phenotypes in pulmonary fibrosis. Am J Respir Cell Mol Biol 2003;29:S87–S92. - PubMed
-
- Powell DW, Mifflin RC, Valentich JD, Crowe SE, Saada JI, West AB. Myofibroblasts: I. Paracrine cells important in health and disease. Am J Physiol 1999;277:C1–C9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

