Midkine translocated to nucleoli and involved in carcinogenesis
- PMID: 19152444
- PMCID: PMC2653361
- DOI: 10.3748/wjg.15.412
Midkine translocated to nucleoli and involved in carcinogenesis
Abstract
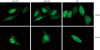
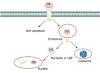
Midkine (MK) is a heparin-binding growth factor with its gene first identified in embryonal carcinoma cells at early stages of retinoic acid-induced differentiation. MK is frequently and highly expressed in a variety of human carcinomas. Furthermore, the blood MK level is frequently elevated with advance of human carcinomas, decreased after surgical removal of the tumors. Thus, it is expected to become a promising marker for evaluating the progress of carcinomas. There is mounting evidence that MK plays a significant role in carcinogenesis-related activities, such as proliferation, migration, anti-apoptosis, mitogenesis, transforming, and angiogenesis. In addition, siRNA and anti-sense oligonucleotides for MK have yielded great effects in anti-tumor activities. Therefore, MK appears to be a potential candidate molecular target of therapy for human carcinomas. In this paper, we review MK targeting at nucleoli in different tumor cells and its role in carcinogenesis to deepen our understanding of the mechanism of MK involved in carcinogenesis.
Figures

Similar articles
-
Midkine accumulated in nucleolus of HepG2 cells involved in rRNA transcription.World J Gastroenterol. 2008 Oct 28;14(40):6249-53. doi: 10.3748/wjg.14.6249. World J Gastroenterol. 2008. PMID: 18985819 Free PMC article.
-
Midkine and pleiotrophin in neural development and cancer.Cancer Lett. 2004 Feb 20;204(2):127-43. doi: 10.1016/S0304-3835(03)00450-6. Cancer Lett. 2004. PMID: 15013213 Review.
-
Construction of a fusion protein expression vector MK-EGFP and its subcellular localization in different carcinoma cell lines.World J Gastroenterol. 2006 Dec 21;12(47):7649-53. doi: 10.3748/wjg.v12.i47.7649. World J Gastroenterol. 2006. PMID: 17171794 Free PMC article.
-
Antisense oligonucleotides targeting midkine induced apoptosis and increased chemosensitivity in hepatocellular carcinoma cells.Acta Pharmacol Sin. 2006 Dec;27(12):1630-6. doi: 10.1111/j.1745-7254.2006.00459.x. Acta Pharmacol Sin. 2006. Retraction in: Acta Pharmacol Sin. 2019 Nov;40(11):1501. doi: 10.1038/s41401-019-0235-7. PMID: 17112419 Retracted.
-
The midkine family in cancer, inflammation and neural development.Nagoya J Med Sci. 2005 Jun;67(3-4):71-82. Nagoya J Med Sci. 2005. PMID: 17375473 Review.
Cited by
-
Midkine upregulates MICA/B expression in human gastric cancer cells and decreases natural killer cell cytotoxicity.Cancer Immunol Immunother. 2012 Oct;61(10):1745-53. doi: 10.1007/s00262-012-1235-3. Epub 2012 Mar 14. Cancer Immunol Immunother. 2012. PMID: 22415659 Free PMC article.
-
Comparison of diagnostic accuracy of Midkine and AFP for detecting hepatocellular carcinoma: a systematic review and meta-analysis.Biosci Rep. 2020 Mar 27;40(3):BSR20192424. doi: 10.1042/BSR20192424. Biosci Rep. 2020. PMID: 32039435 Free PMC article.
-
Diagnostic and Prognostic Value of Talin-1 and Midkine as Tumor Markers in Hepatocellular Carcinoma in Egyptian Patients.Asian Pac J Cancer Prev. 2018 Jun 25;19(6):1503-1508. doi: 10.22034/APJCP.2018.19.6.1503. Asian Pac J Cancer Prev. 2018. PMID: 29936723 Free PMC article.
-
Deciphering GRINA/Lifeguard1: Nuclear Location, Ca2+ Homeostasis and Vesicle Transport.Int J Mol Sci. 2019 Aug 16;20(16):4005. doi: 10.3390/ijms20164005. Int J Mol Sci. 2019. PMID: 31426446 Free PMC article. Review.
-
Midkine noncanonically suppresses AMPK activation through disrupting the LKB1-STRAD-Mo25 complex.Cell Death Dis. 2022 Apr 29;13(4):414. doi: 10.1038/s41419-022-04801-0. Cell Death Dis. 2022. PMID: 35487917 Free PMC article.
References
-
- Muramatsu T. Midkine and pleiotrophin: two related proteins involved in development, survival, inflammation and tumorigenesis. J Biochem. 2002;132:359–371. - PubMed
-
- Nakagawara A, Milbrandt J, Muramatsu T, Deuel TF, Zhao H, Cnaan A, Brodeur GM. Differential expression of pleiotrophin and midkine in advanced neuroblastomas. Cancer Res. 1995;55:1792–1797. - PubMed
-
- Tsutsui J, Kadomatsu K, Matsubara S, Nakagawara A, Hamanoue M, Takao S, Shimazu H, Ohi Y, Muramatsu T. A new family of heparin-binding growth/differentiation factors: increased midkine expression in Wilms’ tumor and other human carcinomas. Cancer Res. 1993;53:1281–1285. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

