Pharmacological characterization of recombinant NR1/NR2A NMDA receptors with truncated and deleted carboxy termini expressed in Xenopus laevis oocytes
- PMID: 19154422
- PMCID: PMC2697674
- DOI: 10.1111/j.1476-5381.2008.00040.x
Pharmacological characterization of recombinant NR1/NR2A NMDA receptors with truncated and deleted carboxy termini expressed in Xenopus laevis oocytes
Abstract
Background and purpose: The carboxy terminal domain (CTD) of NR2 N-methyl-d-aspartate receptor (NMDAR) subunits interacts with numerous scaffolding and signal transduction proteins. Mutations of this region affect trafficking and downstream signalling of NMDARs. This study determines to what extent characteristic pharmacological properties of NR2A-containing NMDARs are influenced by this key functional domain.
Experimental approach: Using recombinant receptor expression in Xenopus laevis oocytes and two electrode voltage clamp recordings we characterized pharmacological properties of rat NR1/NR2A NMDARs with altered CTDs. We assessed the effects of truncating [at residue Iso1098; NR2A(trunC)] and deleting [from residue Phe822; NR2A(delC)] the CTD of NR2A NMDAR subunits on agonist potencies, channel block by Mg(2+) and memantine and potentiation of NMDAR-mediated responses by chelating contaminating divalent cations.
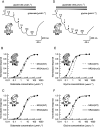
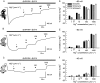
Key results: Truncation or deletion of the CTD of NR2A NMDAR subunits did not affect glutamate potency [EC(50) = 2.2 micromol.L(-1), NR2A(trunC); 2.7 micromol.L(-1), NR2A(delC) compared with 3.3 micromol.L(-1), NR2A(WT)] but did significantly increase glycine potency [EC(50) = 500 nmol.L(-1), NR2A(trunC); 900 nmol.L(-1), NR2A(delC) compared with 1.3 micromol.L(-1), NR2A(WT)]. Voltage-dependent Mg(2+) block of NR2A(WT)- and NR2A(trunC)-containing NMDARs was similar but low concentrations of Mg(2+) (1 micromol.L(-1)) potentiated NR1/NR2A(delC) NMDARs. Memantine block was not affected by changes to the structure of the NR2A CTD. EDTA-induced potentiation was similar at each of the three NMDAR constructs.
Conclusions and implications: Of the parameters studied only minor influences of the CTD were observed; these are unlikely to compromise interpretation of studies that make use of CTD-mutated recombinant receptors or transgenic mice in investigations of the role of the CTD in NMDAR signalling.
Figures



Similar articles
-
Mg2+ and memantine block of rat recombinant NMDA receptors containing chimeric NR2A/2D subunits expressed in Xenopus laevis oocytes.J Physiol. 2008 Jan 1;586(1):211-25. doi: 10.1113/jphysiol.2007.143164. Epub 2007 Oct 25. J Physiol. 2008. PMID: 17962329 Free PMC article.
-
Modulation of glycine potency in rat recombinant NMDA receptors containing chimeric NR2A/2D subunits expressed in Xenopus laevis oocytes.J Physiol. 2008 Jan 1;586(1):227-45. doi: 10.1113/jphysiol.2007.143172. Epub 2007 Oct 25. J Physiol. 2008. PMID: 17962328 Free PMC article.
-
The serine protease plasmin cleaves the amino-terminal domain of the NR2A subunit to relieve zinc inhibition of the N-methyl-D-aspartate receptors.J Biol Chem. 2009 May 8;284(19):12862-73. doi: 10.1074/jbc.M805123200. Epub 2009 Feb 24. J Biol Chem. 2009. PMID: 19240037 Free PMC article.
-
N-[2-(N-(2-mercaptoethyl)) amino ethyl]-N-(2-mercaptoethyl)-3,5-dimethylacetamide amantadine-technetium.2012 Jun 12 [updated 2012 Jul 17]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2012 Jun 12 [updated 2012 Jul 17]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22812023 Free Books & Documents. Review.
-
Influence of the NR3A subunit on NMDA receptor functions.Prog Neurobiol. 2010 May;91(1):23-37. doi: 10.1016/j.pneurobio.2010.01.004. Epub 2010 Jan 25. Prog Neurobiol. 2010. PMID: 20097255 Free PMC article. Review.
Cited by
-
d-Serine Inhibits Non-ionotropic NMDA Receptor Signaling.J Neurosci. 2024 Aug 7;44(32):e0140242024. doi: 10.1523/JNEUROSCI.0140-24.2024. J Neurosci. 2024. PMID: 38942470 Free PMC article.
-
Structure, Function, and Pharmacology of Glutamate Receptor Ion Channels.Pharmacol Rev. 2021 Oct;73(4):298-487. doi: 10.1124/pharmrev.120.000131. Pharmacol Rev. 2021. PMID: 34753794 Free PMC article. Review.
-
Disease-associated nonsense and frame-shift variants resulting in the truncation of the GluN2A or GluN2B C-terminal domain decrease NMDAR surface expression and reduce potentiating effects of neurosteroids.Cell Mol Life Sci. 2024 Jan 12;81(1):36. doi: 10.1007/s00018-023-05062-6. Cell Mol Life Sci. 2024. PMID: 38214768 Free PMC article.
-
The NMDA receptor intracellular C-terminal domains reciprocally interact with allosteric modulators.Biochem Pharmacol. 2019 Jan;159:140-153. doi: 10.1016/j.bcp.2018.11.018. Epub 2018 Nov 29. Biochem Pharmacol. 2019. PMID: 30503374 Free PMC article.
-
Amino terminal domains of the NMDA receptor are organized as local heterodimers.PLoS One. 2011 Apr 22;6(4):e19180. doi: 10.1371/journal.pone.0019180. PLoS One. 2011. PMID: 21544205 Free PMC article.
References
-
- Burnashev N, Schoepfer R, Monyer H, Ruppersberg JP, Gunter W, Seeburg PH, et al. Control by asparagine residues of calcium permeability and magnesium blockade in the NMDA receptor. Science. 1992;257:1415–1419. - PubMed
-
- Chen HSV, Lipton SA. Pharmacological implications of two distinct mechanisms of interaction of memantine with N-methyl-d-aspartate-gated channels. J Pharmacol Exp Ther. 2005;314:961–971. - PubMed
-
- Chen PE, Geballe MT, Stansfeld PJ, Johnston AR, Yuan H, Jacob AL, et al. Structural features of the glutamate binding site in recombinant NR1/NR2A N-methyl-d-aspartate receptors determined by site-directed mutagenesis and molecular modeling. Mol Pharmacol. 2005;67:1470–1484. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

