IFN regulatory factor 8 sensitizes soft tissue sarcoma cells to death receptor-initiated apoptosis via repression of FLICE-like protein expression
- PMID: 19155307
- PMCID: PMC2633427
- DOI: 10.1158/0008-5472.CAN-08-2520
IFN regulatory factor 8 sensitizes soft tissue sarcoma cells to death receptor-initiated apoptosis via repression of FLICE-like protein expression
Abstract
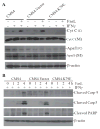
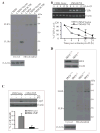
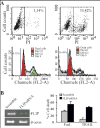
IFN regulatory factor 8 (IRF8) has been shown to suppress tumor development at least partly through regulating apoptosis of tumor cells; however, the molecular mechanisms underlying IRF8 regulation of apoptosis are still not fully understood. Here, we showed that disrupting IRF8 function resulted in inhibition of cytochrome c release, caspase-9 and caspase-3 activation, and poly(ADP-ribose) polymerase cleavage in soft tissue sarcoma (STS) cells. Inhibition of the mitochondrion-dependent apoptosis signaling cascade is apparently due to blockage of caspase-8 and Bid activation. Analysis of signaling events upstream of caspase-8 revealed that disrupting IRF8 function dramatically increases FLIP mRNA stability, resulting in increased IRF8 protein level. Furthermore, primary myeloid cells isolated from IRF8-null mice also exhibited increased FLIP protein level, suggesting that IRF8 might be a general repressor of FLIP. Nuclear IRF8 protein was absent in 92% (55 of 60) of human STS specimens, and 99% (59 of 60) of human STS specimens exhibited FLIP expression, suggesting that the nuclear IRF8 protein level is inversely correlated with FLIP level in vivo. Silencing FLIP expression significantly increased human sarcoma cells to both FasL-induced and tumor necrosis factor-related apoptosis-inducing ligand (TRAIL)-induced apoptosis, and ectopic expression of IRF8 also significantly increased the sensitivity of these human sarcoma cells to FasL- and TRAIL-induced apoptosis. Taken together, our data suggest that IRF8 mediates FLIP expression level to regulate apoptosis and targeting IRF8 expression is a potentially effective therapeutic strategy to sensitize apoptosis-resistant human STS to apoptosis, thereby possibly overcoming chemoresistance of STS, currently a major obstacle in human STS therapy.
Figures



Similar articles
-
α-TEA induces apoptosis of human breast cancer cells via activation of TRAIL/DR5 death receptor pathway.Mol Carcinog. 2010 Nov;49(11):964-73. doi: 10.1002/mc.20681. Mol Carcinog. 2010. PMID: 20886583
-
Selective inhibition of FLICE-like inhibitory protein expression with small interfering RNA oligonucleotides is sufficient to sensitize tumor cells for TRAIL-induced apoptosis.Mol Med. 2002 Nov;8(11):725-32. Mol Med. 2002. PMID: 12520089 Free PMC article.
-
DR5-mediated DISC controls caspase-8 cleavage and initiation of apoptosis in human glioblastomas.J Cell Mol Med. 2010 Jun;14(6A):1303-17. doi: 10.1111/j.1582-4934.2009.00777.x. Epub 2009 May 11. J Cell Mol Med. 2010. PMID: 19432816 Free PMC article.
-
Down-regulation of intracellular anti-apoptotic proteins, particularly c-FLIP by therapeutic agents; the novel view to overcome resistance to TRAIL.J Cell Physiol. 2018 Oct;233(10):6470-6485. doi: 10.1002/jcp.26585. Epub 2018 May 9. J Cell Physiol. 2018. PMID: 29741767 Review.
-
c-FLIP, a master anti-apoptotic regulator.Exp Oncol. 2012 Oct;34(3):176-84. Exp Oncol. 2012. PMID: 23070002 Free PMC article. Review.
Cited by
-
Deregulation of apoptotic factors Bcl-xL and Bax confers apoptotic resistance to myeloid-derived suppressor cells and contributes to their persistence in cancer.J Biol Chem. 2013 Jun 28;288(26):19103-15. doi: 10.1074/jbc.M112.434530. Epub 2013 May 15. J Biol Chem. 2013. PMID: 23677993 Free PMC article.
-
TNFα cooperates with IFN-γ to repress Bcl-xL expression to sensitize metastatic colon carcinoma cells to TRAIL-mediated apoptosis.PLoS One. 2011 Jan 17;6(1):e16241. doi: 10.1371/journal.pone.0016241. PLoS One. 2011. PMID: 21264227 Free PMC article.
-
CXCL16 suppresses liver metastasis of colorectal cancer by promoting TNF-α-induced apoptosis by tumor-associated macrophages.BMC Cancer. 2014 Dec 15;14:949. doi: 10.1186/1471-2407-14-949. BMC Cancer. 2014. PMID: 25495942 Free PMC article.
-
IRF8: Mechanism of Action and Health Implications.Cells. 2022 Aug 24;11(17):2630. doi: 10.3390/cells11172630. Cells. 2022. PMID: 36078039 Free PMC article. Review.
-
NF-κB directly regulates Fas transcription to modulate Fas-mediated apoptosis and tumor suppression.J Biol Chem. 2012 Jul 20;287(30):25530-40. doi: 10.1074/jbc.M112.356279. Epub 2012 Jun 5. J Biol Chem. 2012. PMID: 22669972 Free PMC article.
References
-
- Mocellin S, Rossi CR, Brandes A, Nitti D. Adult soft tissue sarcomas: conventional therapies and molecularly targeted approaches. Cancer Treat Rev. 2006;32:9–27. - PubMed
-
- Kanno Y, Levi BZ, Tamura T, Ozato K. Immune cell-specific amplification of interferon signaling by the IRF-4/8-PU.1 complex. J Interferon Cytokine Res. 2005;25:770–9. - PubMed
-
- Levi BZ, Hashmueli S, Gleit-Kielmanowicz M, Azriel A, Meraro D. ICSBP/IRF-8 transactivation: a tale of protein-protein interaction. J Interferon Cytokine Res. 2002;22:153–60. - PubMed
-
- Holtschke T, Lohler J, Kanno Y, et al. Immunodeficiency and chronic myelogenous leukemia-like syndrome in mice with a targeted mutation of the ICSBP gene. Cell. 1996;87:307–17. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

