Insulin-like growth factor-1 promotes G(1)/S cell cycle progression through bidirectional regulation of cyclins and cyclin-dependent kinase inhibitors via the phosphatidylinositol 3-kinase/Akt pathway in developing rat cerebral cortex
- PMID: 19158303
- PMCID: PMC3256126
- DOI: 10.1523/JNEUROSCI.1700-08.2009
Insulin-like growth factor-1 promotes G(1)/S cell cycle progression through bidirectional regulation of cyclins and cyclin-dependent kinase inhibitors via the phosphatidylinositol 3-kinase/Akt pathway in developing rat cerebral cortex
Abstract
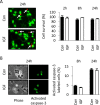
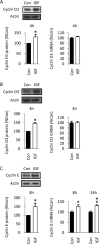
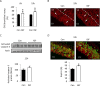
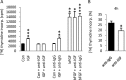
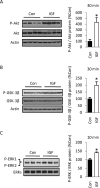
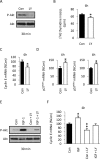
Although survival-promoting effects of insulin-like growth factor-1 (IGF-1) during neurogenesis are well characterized, mitogenic effects remain less well substantiated. Here, we characterize cell cycle regulators and signaling pathways underlying IGF-1 effects on embryonic cortical precursor proliferation in vitro and in vivo. In vitro, IGF-1 stimulated cell cycle progression and increased cell number without promoting cell survival. IGF-1 induced rapid increases in cyclin D1 and D3 protein levels at 4 h and cyclin E at 8 h. Moreover, p27(KIP1) and p57(KIP2) expression were reduced, suggesting downregulation of negative regulators contributes to mitogenesis. Furthermore, the phosphatidylinositol 3-kinase (PI3K)/Akt pathway specifically underlies IGF-1 activity, because blocking this pathway, but not MEK (mitogen-activated protein kinase kinase)/ERK (extracellular signal-regulated kinase), prevented mitogenesis. To determine whether mechanisms defined in culture relate to corticogenesis in vivo, we performed transuterine intracerebroventricular injections. Whereas blockade of endogenous factor with anti-IGF-1 antibody decreased DNA synthesis, IGF-1 injection stimulated DNA synthesis and increased the number of S-phase cells in the ventricular zone. IGF-1 treatment increased phospho-Akt fourfold at 30 min, cyclins D1 and E by 6 h, and decreased p27(KIP1) and p57(KIP2) expression. Moreover, blockade of the PI3K/Akt pathway in vivo decreased DNA synthesis and cyclin E, increased p27(KIP1) and p57(KIP2) expression, and prevented IGF-1-induced cyclin E mRNA upregulation. Finally, IGF-1 injection in embryos increased postnatal day 10 brain DNA content by 28%, suggesting a role for IGF-1 in brain growth control. These results demonstrate a mitogenic role for IGF-1 that tightly controls both positive and negative cell cycle regulators, and indicate that the PI3K/Akt pathway mediates IGF-1 mitogenic signaling during corticogenesis.
Figures





References
-
- Aberg MA, Aberg ND, Palmer TD, Alborn AM, Carlsson-Skwirut C, Bang P, Rosengren LE, Olsson T, Gage FH, Eriksson PS. IGF-I has a direct proliferative effect in adult hippocampal progenitor cells. Mol Cell Neurosci. 2003;24:23–40. - PubMed
-
- Aizenman Y, de Vellis J. Brain neurons develop in a serum and glial free environment: effects of transferrin, insulin, insulin-like growth factor-I and thyroid hormone on neuronal survival, growth and differentiation. Brain Res. 1987;406:32–42. - PubMed
-
- Beck KD, Powell-Braxton L, Widmer HR, Valverde J, Hefti F. Igf1 gene disruption results in reduced brain size, CNS hypomyelination, and loss of hippocampal granule and striatal parvalbumin-containing neurons. Neuron. 1995;14:717–730. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous
