Long-term safety and tolerability of rivastigmine in patients with Alzheimer's disease switched from donepezil: an open-label extension study
- PMID: 19158974
- PMCID: PMC2629056
- DOI: 10.4088/pcc.v10n0503
Long-term safety and tolerability of rivastigmine in patients with Alzheimer's disease switched from donepezil: an open-label extension study
Abstract
Objectives: The objective of this article is to present safety and tolerability data from the long-term extension phase of a core study conducted in patients with Alzheimer's disease (AD) who were immediately switched to rivastigmine.
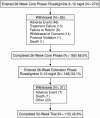
Method: This was a 26-week open-label extension (OLE) of a prospective, 26-week, open-label, single-arm, multicenter study conducted in the United States from October 2003 to January 2005. Patients had a diagnosis of Alzheimer's disease according to DSM-IV-TR and National Institute of Neurologic and Communicative Disorders and Stroke and the Alzheimer's Disease and Related Disorders Association criteria. Safety and tolerability of rivastigmine were monitored through monthly telephone contacts. At week 52, patients or caregivers were contacted by telephone to evaluate the patient's well-being.
Results: 146 patients (approximately 79% of patients who completed the core phase) entered this OLE. Most patients (N = 115, 78.8%) completed the full 26 weeks of the extension phase, during which time they received a mean rivastigmine dosage of 10.5 mg/day. The number of patients reporting newly occurring or worsening adverse events decreased considerably during the OLE (N = 84, 57.5%) compared with the core phase (the first 26 weeks; N = 116, 79.5%). Most patients reported adverse events that were mild or moderate in severity. At the end of the OLE, the majority of patients (128/146; 87.7%) were still receiving treatment with rivastigmine. At week 52, most caregivers expressed satisfaction with rivastigmine treatment (77.4%) and with the changes observed in the patient's behavior during the study (71.9%).
Conclusions: For patients not tolerating or not responding to donepezil, treatment with rivastigmine was safe and well tolerated for at least 52 weeks.
Similar articles
-
Switching from oral donepezil to rivastigmine transdermal patch in Alzheimer's disease: 20-week extension phase results.Prim Care Companion J Clin Psychiatry. 2010;12(5):PCC.09m00852. doi: 10.4088/PCC.09m00852oli. Prim Care Companion J Clin Psychiatry. 2010. PMID: 21274364 Free PMC article.
-
Safety and efficacy of rivastigmine in patients with Alzheimer's disease not responding adequately to donepezil: an open-label study.Prim Care Companion J Clin Psychiatry. 2008;10(4):291-8. doi: 10.4088/pcc.v10n0404. Prim Care Companion J Clin Psychiatry. 2008. PMID: 18787673 Free PMC article.
-
Donepezil and rivastigmine in the treatment of Alzheimer's disease: a best-evidence synthesis of the published data on their efficacy and cost-effectiveness.Clin Ther. 2002 Jun;24(6):862-86; discussion 837. doi: 10.1016/s0149-2918(02)80004-2. Clin Ther. 2002. PMID: 12117079 Review.
-
A 25-week, open-label trial investigating rivastigmine transdermal patches with concomitant memantine in mild-to-moderate Alzheimer's disease: a post hoc analysis.Curr Med Res Opin. 2010 Feb;26(2):263-9. doi: 10.1185/03007990903434914. Curr Med Res Opin. 2010. PMID: 19929593 Clinical Trial.
-
Treatment of Alzheimer's disease in the long-term-care setting.Am J Health Syst Pharm. 2009 May 15;66(10):899-907. doi: 10.2146/ajhp070622. Am J Health Syst Pharm. 2009. PMID: 19420308 Review.
Cited by
-
Pharmacokinetic Drug-Drug Interaction and Responsible Mechanism between Memantine and Cimetidine.Pharmaceutics. 2018 Aug 6;10(3):119. doi: 10.3390/pharmaceutics10030119. Pharmaceutics. 2018. PMID: 30082658 Free PMC article.
References
-
- Small GW, Rabins PV, Barry PP, et al. Diagnosis and treatment of Alzheimer disease and related disorders: consensus statement of the American Association for Geriatric Psychiatry, the Alzheimer's Association, and the American Geriatrics Society. JAMA. 1997;278:1363–1371. - PubMed
-
- Kawas C, Gray S, Brookmeyer R, et al. Age-specific incidence rates of Alzheimer's disease: the Baltimore Longitudinal Study of Aging. Neurology. 2000;54:2072–2077. - PubMed
-
- Jorm AF, Jolley D. The incidence of dementia: a meta-analysis. Neurology. 1998;51:728–733. - PubMed
-
- Guttman R, Altman RD, Nielsen NH. Alzheimer disease: report of the Council on Scientific Affairs. Arch Fam Med. 1999;8:347–353. - PubMed
-
- Trabucchi M. An economic perspective on Alzheimer's disease. J Geriatr Psychiatry Neurol. 1999;12:29–38. - PubMed
LinkOut - more resources
Full Text Sources