Involvement of Th17 cells and the effect of anti-IL-6 therapy in autoimmune uveitis
- PMID: 19164426
- PMCID: PMC2722800
- DOI: 10.1093/rheumatology/ken489
Involvement of Th17 cells and the effect of anti-IL-6 therapy in autoimmune uveitis
Abstract
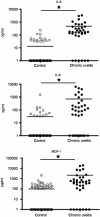
Objectives: Human endogenous uveitis is one of the sight-threatening diseases associated with variety of systemic disorders, such as Behcet's disease and sarcoidosis. Recently, biosynthesized antibodies against inflammatory cytokines have been recognized to be useful to control the regional inflammation. In this study, we focused on the possibility of IL-6-based biological therapies for endogenous uveitis. We initially confirmed the significant increase of several inflammatory soluble factors including IL-6 in the vitreous fluids from refractory/chronic engogenous uveitis patients.
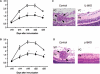
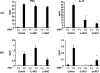
Methods: To investigate the role of IL-6 in the formation of refractory ocular inflammation, we used the mouse experimental autoimmune uveitis (EAU) model. Both IL-6 and IL-23 are required for the development of IL-17-producing helper T subset (Th17) from naïve CD4(+) T cells. Results. In the EAU model, neither IL-6-deficient mice nor IL-23-deficient mice could induce Th17 cells and the EAU score was decreased in these mice in the entire time course. We also confirmed that systemic administration of anti-il-6 receptor antibody ameliorates EAU By suppressing both systemic and regional TH17 responses.
Conclusions: IL-6 is responsible for causing ocular inflammation, and it is, at least partially, due to IL-6-dependent Th17 differentiation. IL-6 may be a target for therapy of refractory endogenous uveitis in humans.
Figures


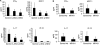
Similar articles
-
Blockade of interleukin-6 signaling suppresses experimental autoimmune uveoretinitis by the inhibition of inflammatory Th17 responses.Exp Eye Res. 2010 Aug;91(2):162-70. doi: 10.1016/j.exer.2010.04.009. Epub 2010 Apr 24. Exp Eye Res. 2010. PMID: 20420831
-
CTLA4-Ig suppresses development of experimental autoimmune uveitis in the induction and effector phases: Comparison with blockade of interleukin-6.Exp Eye Res. 2015 Nov;140:53-64. doi: 10.1016/j.exer.2015.08.012. Epub 2015 Aug 20. Exp Eye Res. 2015. PMID: 26297802
-
Either a Th17 or a Th1 effector response can drive autoimmunity: conditions of disease induction affect dominant effector category.J Exp Med. 2008 Apr 14;205(4):799-810. doi: 10.1084/jem.20071258. Epub 2008 Apr 7. J Exp Med. 2008. PMID: 18391061 Free PMC article.
-
[Translational research with experimental autoimmune uveoretinitis (EAU)].Nippon Ganka Gakkai Zasshi. 2007 Mar;111(3):137-58; discussion 159. Nippon Ganka Gakkai Zasshi. 2007. PMID: 17402560 Review. Japanese.
-
Targeting Interleukin-23 in the Treatment of Noninfectious Uveitis.Ophthalmology. 2018 Dec;125(12):1977-1983. doi: 10.1016/j.ophtha.2018.05.014. Epub 2018 Jul 4. Ophthalmology. 2018. PMID: 30458922 Free PMC article. Review.
Cited by
-
Cytokines that Modulate the Differentiation of Th17 Cells in Autoimmune Uveitis.J Immunol Res. 2021 Mar 16;2021:6693542. doi: 10.1155/2021/6693542. eCollection 2021. J Immunol Res. 2021. PMID: 33816637 Free PMC article. Review.
-
miR-23a, miR-146a and miR-301a confer predisposition to Vogt-Koyanagi-Harada syndrome but not to Behcet's disease.Sci Rep. 2016 Jan 28;6:20057. doi: 10.1038/srep20057. Sci Rep. 2016. PMID: 26818976 Free PMC article.
-
Tocilizumab treatment for refractory uveitis-related cystoid macular edema.Graefes Arch Clin Exp Ophthalmol. 2013 Nov;251(11):2627-32. doi: 10.1007/s00417-013-2436-y. Epub 2013 Jul 27. Graefes Arch Clin Exp Ophthalmol. 2013. PMID: 23893042
-
Amelioration of experimental autoimmune uveitis by leflunomide in Lewis rats.PLoS One. 2013 Apr 23;8(4):e62071. doi: 10.1371/journal.pone.0062071. Print 2013. PLoS One. 2013. PMID: 23626769 Free PMC article.
-
Interleukin-6 inhibition in the management of non-infectious uveitis and beyond.J Ophthalmic Inflamm Infect. 2019 Sep 16;9(1):17. doi: 10.1186/s12348-019-0182-y. J Ophthalmic Inflamm Infect. 2019. PMID: 31523783 Free PMC article. Review.
References
-
- Ohno S, Nakamura S, Hori S, et al. Efficacy, safety, and pharmacokinetics of multiple administration of infliximab in Behcet's disease with refractory uveoretinitis. J Rheumatol. 2004;31:1362–8. - PubMed
-
- Imrie FR, Dick AD. Biologics in the treatment of uveitis. Curr Opin Ophthalmol. 2007;18:481–6. - PubMed
-
- Ackermann C, Kavanaugh A. Tumor necrosis factor as a therapeutic target of rheumatologic disease. Expert Opin Ther Targets. 2007;11:1369–84. - PubMed
-
- McLeod C, Bagust A, Boland A, et al. Adalimumab, etanercept and infliximab for the treatment of ankylosing spondylitis: a systematic review and economic evaluation. Health Technol Assess. 2007;11:1–158. iii–iv. - PubMed
-
- Veres G, Baldassano RN, Mamula P. Infliximab therapy for pediatric Crohn's disease. Expert Opin Biol Ther. 2007;7:1869–80. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

