Pneumonic plague pathogenesis and immunity in Brown Norway rats
- PMID: 19164505
- PMCID: PMC2665751
- DOI: 10.2353/ajpath.2009.071168
Pneumonic plague pathogenesis and immunity in Brown Norway rats
Abstract
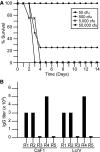
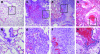
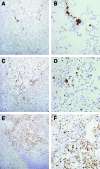
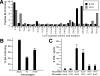
The Brown Norway rat was recently described as a bubonic plague model that closely mimics human disease. We therefore evaluated the Brown Norway rat as an alternative small animal model for pneumonic plague and characterized both the efficacy and potency of vaccine candidates. When infected by intranasal instillation, these rats rapidly developed fatal pneumonic plague within 2 to 4 days of infection. Plague disease was characterized by severe alveolar edema and vascular hemorrhage in the lung in addition to fulminant necrotizing pneumonia caused by massive bacterial replication and inflammation. Twenty-four hours before death, animals developed systemic disease with an apparent delayed inflammatory response. We evaluated the ability of the protective antigen, LcrV, and a mutant derivative, V10, to protect these rats from pneumonic plague. Both were highly effective vaccines because complete protection was observed at challenge doses of 7500 LD(50). Antibody analyses suggested stronger potency of V10 immune sera compared with LcrV in the passive transfer of immunity to bubonic plague, with multiple neutralizing epitopes in LcrV. Taken together, these data demonstrate the effectiveness of inhibiting type III secretion in the prevention of pneumonic plague in rats and reveal critical contributions from both the cellular and humoral immune systems. Thus, the Brown Norway rat is an appealing alternative small animal model for the study of pneumonic plague pathogenesis and immunity.
Figures



References
-
- Lien-Teh W. Geneva: League of Nations Health Organization,; A Treatise on Pneumonic Plague. 1926
-
- Pollitzer R. Geneva: World Health Organization,; Plague. 1954 - PubMed
-
- Gage K, Kosoy M. Natural history of plague: perspectives from more than a century of research. Annu Rev Entomol. 2005;50:505–528. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

