The contribution of synaptic inputs to sustained depolarizations in reticulospinal neurons
- PMID: 19176823
- PMCID: PMC6665139
- DOI: 10.1523/JNEUROSCI.3073-08.2009
The contribution of synaptic inputs to sustained depolarizations in reticulospinal neurons
Abstract
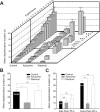
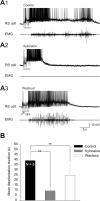
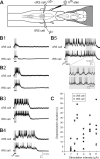
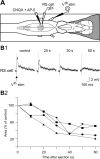
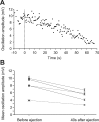
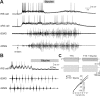
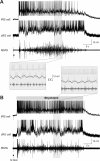
Sensory stimulation elicits sustained depolarizations in lamprey reticulospinal (RS) cells for which intrinsic properties were shown to play a crucial role. The depolarizations last up to minutes, and we tested whether the intrinsic properties required the cooperation of synaptic inputs to maintain RS cells depolarized for such long periods of time. Ascending spinal inputs to RS cells were reversibly blocked by applying xylocaine over the rostral spinal cord segments. The duration of the sustained depolarizations was markedly reduced. The membrane potential oscillations in tune with locomotor activity that were present under control condition were also abolished. The contribution of excitatory glutamatergic inputs was then assessed by applying CNQX and AP-5 over one of two simultaneously recorded homologous RS cells on each side of the brainstem. The level of sensory-evoked depolarization decreased significantly in the cell exposed to the antagonists compared with the other RS cell monitored as a control. In contrast, local application of glycine only produced a transient membrane potential hyperpolarization with a marked reduction in the amplitude of membrane potential oscillations. Locally applied strychnine did not change the duration of the sustained depolarizations, suggesting that mechanisms other than glycinergic inhibition are involved in ending the sustained depolarizations in RS cells. It is concluded that excitatory glutamatergic inputs, including ascending spinal feedback, cooperate with intrinsic properties of RS cells to maintain the cells depolarized for prolonged periods, sustaining long bouts of escape swimming.
Figures



References
-
- Alford S, Dubuc R. Glutamate metabotropic receptor mediated depression of synaptic inputs to lamprey reticulospinal neurones. Brain Res. 1993;605:175–179. - PubMed
-
- Brocard F, Dubuc R. Differential contribution of reticulospinal cells to the control of locomotion induced by the mesencephalic locomotor region. J Neurophysiol. 2003;90:1714–1727. - PubMed
-
- Buchanan JT. Identification of interneurons with contralateral, caudal axons in the lamprey spinal cord: synaptic interactions and morphology. J Neurophysiol. 1982;47:961–975. - PubMed
-
- Buchanan JT. The roles of spinal interneurons and motoneurons in the lamprey locomotor network. Prog Brain Res. 1999;123:311–321. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
