High-resolution NMR characterization of a spider-silk mimetic composed of 15 tandem repeats and a CRGD motif
- PMID: 19177364
- PMCID: PMC2708025
- DOI: 10.1002/pro.12
High-resolution NMR characterization of a spider-silk mimetic composed of 15 tandem repeats and a CRGD motif
Abstract
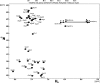
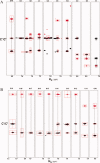
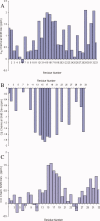
Multidimensional solution NMR spectroscopic techniques have been used to obtain atomic level information about a recombinant spider silk construct in hexafluoro-isopropanol (HFIP). The synthetic 49 kDa silk-like protein mimics authentic silk from Nephila clavipes, with the inclusion of an extracellular matrix recognition motif. 2D (1)H-(15)N HSQC NMR spectroscopy reveals 33 cross peaks, which were assigned to amino acid residues in the semicrystalline repeat units. Signals from the amorphous segments in the primary sequence were weak and broad, suggesting that this region is highly dynamic and undergoing conformational exchange. An analysis of the deviations of the (13)C(alpha), (13)C(beta), and (13)CO chemical shifts relative to the expected random coil values reveals two highly alpha-helical regions from amino acid 12-19 and 26-32, which comprise the polyalanine track and a GGLGSQ sequence. This finding is further supported by phi-value analysis and sequential and medium-range NOE interactions. Pulsed field gradient NMR measurements indicate that the topology of the silk mimetic in HFIP is nonglobular. Moreover, the 3D (15)N-NOESY HSQC spectrum exhibits few long-range NOEs. Similar spectral features have been observed for repeat modules in other polypeptides and are characteristic of an elongated conformation. The results provide a residue-specific description of a silk sequence in nonaqueous solution and may be insightful for understanding the fold and topology of highly concentrated, stable silk before spinning. Additionally, the insights obtained may find application in future design and large-scale production and storage of synthetic silks in organic solvents.
Figures
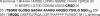


Similar articles
-
Folding recombinant spider-silk in H2 O: Effect of osmolytes on the solution conformation of a 15-repeat spider-silk mimetic.Protein Sci. 2016 Oct;25(10):1853-62. doi: 10.1002/pro.2995. Epub 2016 Aug 19. Protein Sci. 2016. PMID: 27488926 Free PMC article.
-
WISE NMR characterization of nanoscale heterogeneity and mobility in supercontracted Nephila clavipes spider dragline silk.J Am Chem Soc. 2004 May 12;126(18):5867-72. doi: 10.1021/ja031930w. J Am Chem Soc. 2004. PMID: 15125679
-
Quantitative Correlation between the protein primary sequences and secondary structures in spider dragline silks.Biomacromolecules. 2010 Jan 11;11(1):192-200. doi: 10.1021/bm9010672. Biomacromolecules. 2010. PMID: 20000730 Free PMC article.
-
Biotechnological production of spider-silk proteins enables new applications.Macromol Biosci. 2007 Apr 10;7(4):401-9. doi: 10.1002/mabi.200600255. Macromol Biosci. 2007. PMID: 17429812 Review.
-
Spider silks and their applications.Trends Biotechnol. 2008 May;26(5):244-51. doi: 10.1016/j.tibtech.2008.02.006. Epub 2008 Mar 25. Trends Biotechnol. 2008. PMID: 18367277 Review.
Cited by
-
Folding recombinant spider-silk in H2 O: Effect of osmolytes on the solution conformation of a 15-repeat spider-silk mimetic.Protein Sci. 2016 Oct;25(10):1853-62. doi: 10.1002/pro.2995. Epub 2016 Aug 19. Protein Sci. 2016. PMID: 27488926 Free PMC article.
References
-
- Gosline JM, Guerette PA, Ortlepp CS, Savage KN. The mechanical design of spider silks: from fibroin sequence to mechanical function. J Exp Biol. 1999;202:3295–3303. - PubMed
-
- Vollrath F, Knight DP. Liquid crystalline spinning of spider silk. Nature. 2001;410:541–548. - PubMed
-
- Sofia S, McCarthy MB, Gronowicz G, Kaplan DL. Functionalized silk-based biomaterials for bone formation. J Biomed Mater Res. 2001;54:139–148. - PubMed
-
- Wong Po Foo C, Kaplan DL. Genetic engineering of fibrous proteins: spider dragline silk and collagen. Adv Drug Deliv Rev. 2002;54:1131–1143. - PubMed
-
- Bini E, Foo CW, Huang J, Karageorgiou V, Kitchel B, Kaplan DL. RGD-functionalized bioengineered spider dragline silk biomaterial. Biomacromolecules. 2006;7:3139–3145. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

