Hypertensive state, independent of hypertrophy, exhibits an attenuated decrease in systolic function on cardiac kappa-opioid receptor stimulation
- PMID: 19181965
- PMCID: PMC2670690
- DOI: 10.1152/ajpheart.00909.2008
Hypertensive state, independent of hypertrophy, exhibits an attenuated decrease in systolic function on cardiac kappa-opioid receptor stimulation
Abstract
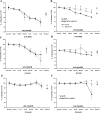
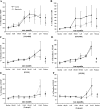
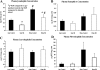
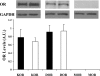
Opioids/opiates are commonly administered to alleviate pain, unload the heart, or decrease breathlessness in patients with advanced heart failure. As such, it is important to evaluate whether the myocardial opioidergic system is altered in cardiac disease. A hamster model of spontaneous hypertension was investigated before the development of hypertension (1 mo of age) and in the hypertensive state (10 mo of age) to evaluate the effect of prolonged hypertension on myocardial opioidergic activity. Plasma beta-endorphin was decreased before the development of hypertension and in the hypertensive state (P < 0.05). There was no change in cardiac beta-endorphin content at either time point. No differences were detected in cardiac or plasma dynorphin A, Met-enkephalin, or Leu-enkephalin, or in cardiac peptide expression of kappa- or delta-opioid receptors. mu-Opioid receptor was not detected in either model. To determine how hypertension affects myocardial opioid signaling, the ex vivo work-performing heart was used to assess the cardiac response to opioid administration in healthy hearts and those subjected to chronic hypertension. Agonists selective for the kappa- and delta-opioid receptors, but not mu-opioid receptors, induced a concentration-dependent decrease in cardiac function. The decrease in left ventricular systolic pressure on administration of the kappa-opioid receptor-selective agonist, U50488H, was attenuated in hearts from hamsters subjected to chronic, untreated hypertension (P < 0.05) compared with control. These results show that peripheral and myocardial opioid expression and signaling are altered in hypertension.
Figures

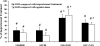
Similar articles
-
Kappa and delta opioid receptor signaling is augmented in the failing heart.J Mol Cell Cardiol. 2009 Oct;47(4):493-503. doi: 10.1016/j.yjmcc.2009.06.016. Epub 2009 Jun 30. J Mol Cell Cardiol. 2009. PMID: 19573531 Free PMC article.
-
The regulation of steroidogenesis by opioid peptides in porcine theca cells.Anim Reprod Sci. 2003 Sep 15;78(1-2):71-84. doi: 10.1016/s0378-4320(03)00042-3. Anim Reprod Sci. 2003. PMID: 12753784
-
Kappa and delta opioid receptor stimulation affects cardiac myocyte function and Ca2+ release from an intracellular pool in myocytes and neurons.Circ Res. 1992 Jan;70(1):66-81. doi: 10.1161/01.res.70.1.66. Circ Res. 1992. PMID: 1309318
-
Involvement of opioid delta (delta)- and kappa (kappa)-receptors in ischemic preconditioning in a rat model of myocardial infarction.Methods Find Exp Clin Pharmacol. 2002 Apr;24(3):139-44. doi: 10.1358/mf.2002.24.3.802298. Methods Find Exp Clin Pharmacol. 2002. PMID: 12087875
-
Altered opioid-mediated control of the spinal release of dynorphin and met-enkephalin in polyarthritic rats.Synapse. 2000 Sep 15;37(4):262-72. doi: 10.1002/1098-2396(20000915)37:4<262::AID-SYN3>3.0.CO;2-6. Synapse. 2000. PMID: 10891863
Cited by
-
Forkhead box F2 regulation of platelet-derived growth factor and myocardin/serum response factor signaling is essential for intestinal development.J Biol Chem. 2015 Mar 20;290(12):7563-75. doi: 10.1074/jbc.M114.609487. Epub 2015 Jan 28. J Biol Chem. 2015. PMID: 25631042 Free PMC article.
-
The Impact of Opium Consumption on Blood Glucose, Serum Lipids and Blood Pressure, and Related Mechanisms.Front Physiol. 2016 Oct 13;7:436. doi: 10.3389/fphys.2016.00436. eCollection 2016. Front Physiol. 2016. PMID: 27790151 Free PMC article. Review.
-
FOXF1 transcription factor promotes lung regeneration after partial pneumonectomy.Sci Rep. 2017 Sep 6;7(1):10690. doi: 10.1038/s41598-017-11175-3. Sci Rep. 2017. PMID: 28878348 Free PMC article.
-
The presence of mu-, delta-, and kappa-opioid receptors in human heart tissue.Heart Vessels. 2014 Nov;29(6):855-63. doi: 10.1007/s00380-013-0456-5. Epub 2014 Jan 5. Heart Vessels. 2014. PMID: 24390763
-
Prospects for Creation of Cardioprotective and Antiarrhythmic Drugs Based on Opioid Receptor Agonists.Med Res Rev. 2016 Sep;36(5):871-923. doi: 10.1002/med.21395. Epub 2016 May 16. Med Res Rev. 2016. PMID: 27197922 Free PMC article. Review.
References
-
- American Heart Association/American Stroke Association. Know the Facts, Get the Stats, 2007. [http://www.americanheart.org/downloadable/heart/116861545709855–1041%20K...].
-
- Barlow MA, Deo S, Johnson S, Caffrey JL. Vagotonic effects of enkephalin are not mediated by sympatholytic mechanisms. Exp Biol Med (Maywood) 231: 387–395, 2006. - PubMed
-
- Barron BA Cardiac opioids. Proc Soc Exp Biol Med 224: 1–7, 2000. - PubMed
-
- Beyer ME, Slesak G, Brehm BR, Hoffmeister HM. Hemodynamic and inotropic effects of the endothelin A antagonist BQ-610 in vivo. J Cardiovasc Pharmacol 31, Suppl 1: S258–S261, 1998. - PubMed
-
- Beyer ME, Slesak G, Hovelborn T, Kazmaier S, Nerz S, Hoffmeister HM. Inotropic effects of endothelin-1: interaction with molsidomine and with BQ 610. Hypertension 33: 145–152, 1999. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

