Mesenchymal stem cell proliferation and differentiation on an injectable calcium phosphate-chitosan composite scaffold
- PMID: 19187958
- PMCID: PMC2662619
- DOI: 10.1016/j.biomaterials.2009.01.022
Mesenchymal stem cell proliferation and differentiation on an injectable calcium phosphate-chitosan composite scaffold
Abstract
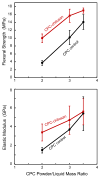
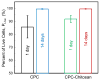
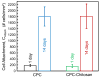
Calcium phosphate cement (CPC) can be molded or injected to form a scaffold in situ, has excellent osteoconductivity, and can be resorbed and replaced by new bone. However, its low strength limits CPC to non-stress-bearing repairs. Chitosan could be used to reinforce CPC, but mesenchymal stem cell (MSC) interactions with CPC-chitosan scaffold have not been examined. The objective of this study was to investigate MSC proliferation and osteogenic differentiation on high-strength CPC-chitosan scaffold. MSCs were harvested from rat bone marrow. At CPC powder/liquid (P/L) mass ratio of 2, flexural strength (mean+/-sd; n=5) was (10.0+/-1.1) MPa for CPC-chitosan, higher than (3.7+/-0.6) MPa for CPC (p<0.05). At P/L of 3, strength was (15.7+/-1.7)MPa for CPC-chitosan, higher than (10.2+/-1.8)MPa for CPC (p<0.05). Percentage of live MSCs attaching to scaffolds increased from 85% at 1 day to 99% at 14 days. There were (180+/-37) cells/mm(2) on scaffold at 1 day; cells proliferated to (1808+/-317) cells/mm(2) at 14 days. SEM showed MSCs with healthy spreading and anchored on nano-apatite crystals via cytoplasmic processes. Alkaline phosphatase activity (ALP) was (557+/-171) (pNPP mM/min)/(microg DNA) for MSCs on CPC-chitosan, higher than (159+/-47) on CPC (p<0.05). Both were higher than (35+/-32) of baseline ALP for undifferentiated MSCs on tissue-culture plastic (p<0.05). In summary, CPC-chitosan scaffold had higher strength than CPC. MSC proliferation on CPC-chitosan matched that of the FDA-approved CPC control. MSCs on the scaffolds differentiated down the osteogenic lineage and expressed high levels of bone marker ALP. Hence, the stronger CPC-chitosan scaffold may be useful for stem cell-based bone regeneration in moderate load-bearing maxillofacial and orthopedic applications.
Figures




References
-
- Laurencin CT, Ambrosio AMA, Borden MD, Cooper JA. Tissue engineering: Orthopedic applications. Annual Rev Biomed Eng. 1999;1:19–46. - PubMed
-
- Hench LL, Polak JM. Third-generation biomedical materials. Science. 2002;295:1014–1017. - PubMed
-
- Praemer A, Furner S, Rice DP. Musculoskeletal conditions in the United States, Foreword. Rosemont: Am Acad Orthop Surg. 1999
-
- Medical Data International. RP-651147 - Orthopedic and musculoskeletal markets: Biotechnology and tissue engineering. 2000:28–31.
-
- Ambrosio AMA, Sahota JS, Khan Y, Laurencin CT. A novel amorphous calcium phosphate polymer ceramic for bone repair: I. Synthesis and characterization. J Biomed Mater Res. 2001;58B:295–301. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

