Scoring diverse cellular morphologies in image-based screens with iterative feedback and machine learning
- PMID: 19188593
- PMCID: PMC2634799
- DOI: 10.1073/pnas.0808843106
Scoring diverse cellular morphologies in image-based screens with iterative feedback and machine learning
Abstract
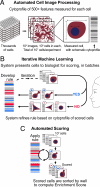
Many biological pathways were first uncovered by identifying mutants with visible phenotypes and by scoring every sample in a screen via tedious and subjective visual inspection. Now, automated image analysis can effectively score many phenotypes. In practical application, customizing an image-analysis algorithm or finding a sufficient number of example cells to train a machine learning algorithm can be infeasible, particularly when positive control samples are not available and the phenotype of interest is rare. Here we present a supervised machine learning approach that uses iterative feedback to readily score multiple subtle and complex morphological phenotypes in high-throughput, image-based screens. First, automated cytological profiling extracts hundreds of numerical descriptors for every cell in every image. Next, the researcher generates a rule (i.e., classifier) to recognize cells with a phenotype of interest during a short, interactive training session using iterative feedback. Finally, all of the cells in the experiment are automatically classified and each sample is scored based on the presence of cells displaying the phenotype. By using this approach, we successfully scored images in RNA interference screens in 2 organisms for the prevalence of 15 diverse cellular morphologies, some of which were previously intractable.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Nusslein-Volhard C, Wieschaus E. Mutations affecting segment number and polarity in Drosophila. Nature. 1980;287:795–801. - PubMed
-
- Morgan TH. The origin of five mutations in eye color in Drosophila and their modes of inheritance. Science. 1911;33:534–537. - PubMed
-
- Muller H. Artificial Transmutation of the Gene. Science. 1927;66:84–87. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

