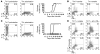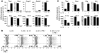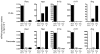Highly purified Th17 cells from BDC2.5NOD mice convert into Th1-like cells in NOD/SCID recipient mice
- PMID: 19188681
- PMCID: PMC2648686
- DOI: 10.1172/JCI37865
Highly purified Th17 cells from BDC2.5NOD mice convert into Th1-like cells in NOD/SCID recipient mice
Abstract
Th17 cells are involved in the pathogenesis of many autoimmune diseases, but it is not clear whether they play a pathogenic role in type 1 diabetes. Here we investigated whether mouse Th17 cells with specificity for an islet antigen can induce diabetes upon transfer into NOD/SCID recipient mice. Induction of diabetes in NOD/SCID mice via adoptive transfer of Th1 cells from BDC2.5 transgenic mice was prevented by treatment of the recipient mice with a neutralizing IFN-γ-specific antibody. This result suggested a major role of Th1 cells in the induction of disease in this model of type 1 diabetes. Nevertheless, transfer of highly purified Th17 cells from BDC2.5 transgenic mice caused diabetes in NOD/SCID recipients with similar rates of onset as in transfer of Th1 cells. However, treatment with neutralizing IL-17-specific antibodies did not prevent disease. Instead, the transferred Th17 cells, completely devoid of IFN-γ at the time of transfer, rapidly converted to secrete IFN-γ in the NOD/SCID recipients. Purified Th17 cells also upregulated Tbet and secreted IFN-γ upon exposure to IL-12 in vitro and in vivo in NOD/SCID recipients. These results indicate substantial plasticity of Th17 commitment toward a Th1-like profile.
Figures