Gene therapy following subretinal AAV5 vector delivery is not affected by a previous intravitreal AAV5 vector administration in the partner eye
- PMID: 19190735
- PMCID: PMC2633462
Gene therapy following subretinal AAV5 vector delivery is not affected by a previous intravitreal AAV5 vector administration in the partner eye
Abstract
Purpose: In an earlier study we found normal adeno-associated viral vector type 2 (AAV2)-mediated GFP expression after intravitreal injection to one eye of normal C57BL/6J mice. However, GFP expression was very poor in the partner eye of the same mouse if this eye received an intravitreal injection of the same vector one month after the initial intravitreal injection. We also found both injections worked well if they were subretinal. In this study, we tested whether the efficiency of subretinal AAV vector transduction is altered by a previous intravitreal injection in the partner eye and more importantly whether therapeutic efficiency is altered in the rd12 mouse (with a recessive RPE65 mutation) after the same injection series.
Methods: One microl of scAAV5-smCBA-GFP (1 x 10(13) genome containing viral particles per ml) was intravitreally injected into the right eyes of four-week-old C57BL/6J mice and 1 microl of scAAV5-smCBA-hRPE65 (1 x 10(13) genome containing viral particles per ml) was intravitreally injected into the right eyes of four-week-old rd12 mice Four weeks later, the same vectors were subretinally injected into the left eyes of the same C57BL/6J and rd12 mice. Left eyes of another cohort of eight-week-old rd12 mice received a single subretinal injection of the same scAAV5-smCBA-hRPE65 vector as the positive control. Dark-adapted electroretinograms (ERGs) were recorded five months after the subretinal injections. AAV-mediated GFP expression in C57BL/6J mice and RPE65 expression and ERG restoration in rd12 mice were evaluated five months after the second subretinal injection. Frozen section analysis was performed for GFP fluorescence in C57BL/6J mice and immunostaining for RPE65 in rd12 eyes.
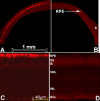
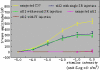
Results: In rd12 mice, dark-adapted ERGs were minimal following the first intravitreal injection of scAAV5-smCBA-RPE65. Following subsequent subretinal injection in the partner eye, dramatic ERG restoration was recorded in that eye. In fact, ERG b-wave amplitudes were statistically similar to those from the eyes that received the initial subretinal injection at a similar age. In C57BL/6J mice, GFP positive cells were detected in eyes following the first intravitreal injection around the injection site. Strong GFP expression in both the retinal pigment epithelium (RPE) and photoreceptor (PR) cells was detected in the partner eyes following the subsequent subretinal injection. Immunostaining of retinal sections with anti-RPE65 antibody showed strong RPE65 expression mainly in the RPE cells of subretinally injected eyes but not in the intravitreally injected eyes except minimally around the injection site.
Conclusions: These results show that an initial intravitreal injection of AAV vectors to one eye of a mouse does not influence AAV-mediated gene expression or related therapeutic effects in the other eye when vectors are administered to the subretinal space. This suggests that the subretinal space possesses a unique immune privilege relative to the vitreous cavity.
Figures


Similar articles
-
Self-complementary AAV5 vector facilitates quicker transgene expression in photoreceptor and retinal pigment epithelial cells of normal mouse.Exp Eye Res. 2010 May;90(5):546-54. doi: 10.1016/j.exer.2010.01.011. Epub 2010 Feb 4. Exp Eye Res. 2010. PMID: 20138034 Free PMC article.
-
Functional and structural recovery of the retina after gene therapy in the RPE65 null mutation dog.Invest Ophthalmol Vis Sci. 2003 Apr;44(4):1663-72. doi: 10.1167/iovs.02-0595. Invest Ophthalmol Vis Sci. 2003. PMID: 12657607
-
Gene therapy rescues cone structure and function in the 3-month-old rd12 mouse: a model for midcourse RPE65 leber congenital amaurosis.Invest Ophthalmol Vis Sci. 2011 Jan 5;52(1):7-15. doi: 10.1167/iovs.10-6138. Print 2011 Jan. Invest Ophthalmol Vis Sci. 2011. PMID: 21169527 Free PMC article.
-
Gene therapy: light is finally in the tunnel.Protein Cell. 2011 Dec;2(12):973-89. doi: 10.1007/s13238-011-1126-y. Epub 2012 Jan 10. Protein Cell. 2011. PMID: 22231356 Free PMC article. Review.
-
Gene therapy of inherited retinopathies: a long and successful road from viral vectors to patients.Hum Gene Ther. 2012 Aug;23(8):796-807. doi: 10.1089/hum.2012.123. Hum Gene Ther. 2012. PMID: 22734691 Free PMC article. Review.
Cited by
-
The frequency-response electroretinogram distinguishes cone and abnormal rod function in rd12 mice.PLoS One. 2015 Feb 23;10(2):e0117570. doi: 10.1371/journal.pone.0117570. eCollection 2015. PLoS One. 2015. PMID: 25706871 Free PMC article.
-
Trans-Corneal Subretinal Injection in Mice and Its Effect on the Function and Morphology of the Retina.PLoS One. 2015 Aug 28;10(8):e0136523. doi: 10.1371/journal.pone.0136523. eCollection 2015. PLoS One. 2015. PMID: 26317758 Free PMC article.
-
rAAV2/5 gene-targeting to rods:dose-dependent efficiency and complications associated with different promoters.Gene Ther. 2010 Sep;17(9):1162-74. doi: 10.1038/gt.2010.56. Epub 2010 Apr 29. Gene Ther. 2010. PMID: 20428215 Free PMC article.
-
Self-complementary AAV-mediated gene therapy restores cone function and prevents cone degeneration in two models of Rpe65 deficiency.Gene Ther. 2010 Jul;17(7):815-26. doi: 10.1038/gt.2010.29. Epub 2010 Mar 18. Gene Ther. 2010. PMID: 20237510 Free PMC article.
-
Intravitreal injection of new adeno-associated viral vector: Enhancing retinoschisin 1 gene transduction in a mouse model of X-linked retinoschisis.Biochem Biophys Rep. 2024 Feb 6;37:101646. doi: 10.1016/j.bbrep.2024.101646. eCollection 2024 Mar. Biochem Biophys Rep. 2024. PMID: 38333050 Free PMC article.
References
-
- Streilein JW. Immune regulation and the eye: a dangerous compromise. FASEB J. 1987;1:199–208. - PubMed
-
- Jiang LQ, Jorquera M, Streilein JW, Ishioka M. Unconventional rejection of neural retinal allografts implanted into the immunologically privileged site of the eye. Transplantation. 1995;59:1201–7. - PubMed
-
- Wenkel H, Streilein JW. Analysis of immune deviation elicited by antigens injected into the subretinal space. Invest Ophthalmol Vis Sci. 1998;39:1823–34. - PubMed
-
- Anand V, Duffy B, Yang ZX, Dejneka NS, Maguire AM, Bennett J. A deviant immune response to viral proteins and transgene product is generated on subretinal administration of adenovirus and adeno-associated virus. Mol Ther. 2002;5:125–32. - PubMed
-
- Blacklow NR, Hoggan MD, Kapikian AZ, Austin JB, Rowe WP. Epidemiology-of adenovirus-associated virus infection in a nursery population. Am J Epidemiol. 1968;88:368–78. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
