Signaling networks that control the lineage commitment and differentiation of bone cells
- PMID: 19191755
- PMCID: PMC3392028
- DOI: 10.1615/critreveukargeneexpr.v19.i1.10
Signaling networks that control the lineage commitment and differentiation of bone cells
Abstract
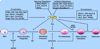
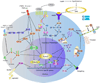
Osteoblasts and osteoclasts are the two major bone cells involved in the bone remodeling process. Osteoblasts are responsible for bone formation while osteoclasts are the bone-resorbing cells. The major event that triggers osteogenesis and bone remodeling is the transition of mesenchymal stem cells into differentiating osteoblast cells and monocyte/macrophage precursors into differentiating osteoclasts. Imbalance in differentiation and function of these two cell types will result in skeletal diseases such as osteoporosis, Paget's disease, rheumatoid arthritis, osteopetrosis, periodontal disease, and bone cancer metastases. Osteoblast and osteoclast commitment and differentiation are controlled by complex activities involving signal transduction and transcriptional regulation of gene expression. Recent advances in molecular and genetic studies using gene targeting in mice enable a better understanding of the multiple factors and signaling networks that control the differentiation process at a molecular level. This review summarizes recent advances in studies of signaling transduction pathways and transcriptional regulation of osteoblast and osteoclast cell lineage commitment and differentiation. Understanding the signaling networks that control the commitment and differentiation of bone cells will not only expand our basic understanding of the molecular mechanisms of skeletal development but will also aid our ability to develop therapeutic means of intervention in skeletal diseases.
Figures


References
-
- Nakamura T, Imai Y, Matsumoto T, Sato S, Takeuchi K, Igarashi K, Harada Y, Azuma Y, Krust A, Yamamoto Y, Nishina H, Takeda S, Takayanagi H, Metzger D, Kanno J, Takaoka K, Martin TJ, Chambon P, Kato S. Estrogen prevents bone loss via estrogen receptor alpha and induction of Fas ligand in osteoclasts. Cell. 2007;130(5):811–823. - PubMed
-
- Stokstad E. Bone quality fills holes in fracture risk. Science. 2005;308(5728):1580–1581. - PubMed
-
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143–147. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

