Human respiratory syncytial virus nonstructural protein NS2 antagonizes the activation of beta interferon transcription by interacting with RIG-I
- PMID: 19193793
- PMCID: PMC2663251
- DOI: 10.1128/JVI.02434-08
Human respiratory syncytial virus nonstructural protein NS2 antagonizes the activation of beta interferon transcription by interacting with RIG-I
Abstract
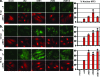
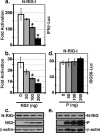
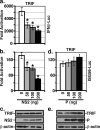
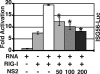
A wide variety of RNA viruses have been shown to produce proteins that inhibit interferon (IFN) production and signaling. For human respiratory syncytial virus (RSV), the nonstructural NS1 and NS2 proteins have been shown to block IFN signaling by causing the proteasomal degradation of STAT2. In addition, recombinant RSVs lacking either NS1 or NS2 induce more IFN production than wild-type (wt) RSV in infected cells. However, the mechanisms by which the NS proteins perform this function are unknown. In this study, we focused on defining the mechanism by which NS2 inhibits the induction of IFN transcription. We find that NS2 is required for the early inhibition of IFN transcription since the infection of cells with NS2-deletion RSV resulted in a higher level of IRF3 activation at early time points postinfection compared with that of wt or NS1-deletion RSV infection. In addition, NS2 expression inhibits IFN transcription induced by both the RIG-I and TLR3 pathways. Furthermore, we show that NS2 inhibits RIG-I-mediated IFN promoter activation by binding to the N-terminal CARD of RIG-I and inhibiting its interaction with the downstream component MAVS (IPS-1, VISA, Cardif). Thus, the RSV NS2 protein is a multifunctional IFN antagonist that targets specific components of both the IFN induction and IFN signaling pathways.
Figures



Similar articles
-
Inhibition of retinoic acid-inducible gene I-mediated induction of beta interferon by the NS1 protein of influenza A virus.J Virol. 2007 Jan;81(2):514-24. doi: 10.1128/JVI.01265-06. Epub 2006 Nov 1. J Virol. 2007. PMID: 17079289 Free PMC article.
-
Hepatitis C virus NS2 and NS3/4A proteins are potent inhibitors of host cell cytokine/chemokine gene expression.Virol J. 2006 Sep 1;3:66. doi: 10.1186/1743-422X-3-66. Virol J. 2006. PMID: 16945160 Free PMC article.
-
Replacement of the respiratory syncytial virus nonstructural proteins NS1 and NS2 by the V protein of parainfluenza virus 5.Virology. 2007 Nov 10;368(1):73-82. doi: 10.1016/j.virol.2007.06.017. Epub 2007 Jul 16. Virology. 2007. PMID: 17632199 Free PMC article.
-
Respiratory syncytial virus nonstructural proteins 1 and 2: Exceptional disrupters of innate immune responses.PLoS Pathog. 2019 Oct 17;15(10):e1007984. doi: 10.1371/journal.ppat.1007984. eCollection 2019 Oct. PLoS Pathog. 2019. PMID: 31622448 Free PMC article. Review.
-
Respiratory syncytial virus mechanisms to interfere with type 1 interferons.Curr Top Microbiol Immunol. 2013;372:173-91. doi: 10.1007/978-3-642-38919-1_9. Curr Top Microbiol Immunol. 2013. PMID: 24362690 Review.
Cited by
-
Respiratory syncytial virus infection modifies and accelerates pulmonary disease via DC activation and migration.J Leukoc Biol. 2013 Jul;94(1):5-15. doi: 10.1189/jlb.0412195. Epub 2013 Jan 4. J Leukoc Biol. 2013. PMID: 23293372 Free PMC article.
-
Innate Immune Components that Regulate the Pathogenesis and Resolution of hRSV and hMPV Infections.Viruses. 2020 Jun 12;12(6):637. doi: 10.3390/v12060637. Viruses. 2020. PMID: 32545470 Free PMC article. Review.
-
Foot-and-Mouth Disease Virus 3B Protein Interacts with Pattern Recognition Receptor RIG-I to Block RIG-I-Mediated Immune Signaling and Inhibit Host Antiviral Response.J Immunol. 2020 Oct 15;205(8):2207-2221. doi: 10.4049/jimmunol.1901333. Epub 2020 Sep 11. J Immunol. 2020. PMID: 32917788 Free PMC article.
-
Human metapneumovirus M2-2 protein inhibits RIG-I signaling by preventing TRIM25-mediated RIG-I ubiquitination.Front Immunol. 2022 Aug 15;13:970750. doi: 10.3389/fimmu.2022.970750. eCollection 2022. Front Immunol. 2022. PMID: 36045682 Free PMC article.
-
Multiple functional domains and complexes of the two nonstructural proteins of human respiratory syncytial virus contribute to interferon suppression and cellular location.J Virol. 2011 Oct;85(19):10090-100. doi: 10.1128/JVI.00413-11. Epub 2011 Jul 27. J Virol. 2011. PMID: 21795342 Free PMC article.
References
-
- Bitko, V., O. Shulyayeva, B. Mazumder, A. Musiyenko, M. Ramaswamy, D. C. Look, and S. Barik. 2007. Nonstructural proteins of respiratory syncytial virus suppress premature apoptosis by an NF-κB-dependent, interferon-independent mechanism and facilitate virus growth. J. Virol. 811786-1795. - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

