O-linked N-acetylglucosaminylation of Sp1 inhibits the human immunodeficiency virus type 1 promoter
- PMID: 19193796
- PMCID: PMC2663287
- DOI: 10.1128/JVI.01384-08
O-linked N-acetylglucosaminylation of Sp1 inhibits the human immunodeficiency virus type 1 promoter
Abstract
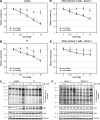
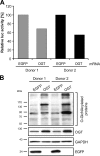
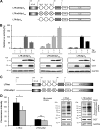
Human immunodeficiency virus type 1 (HIV-1) gene expression and replication are regulated by the promoter/enhancer located in the U3 region of the proviral 5' long terminal repeat (LTR). The binding of cellular transcription factors to specific regulatory sites in the 5' LTR is a key event in the replication cycle of HIV-1. Since transcriptional activity is regulated by the posttranslational modification of transcription factors with the monosaccharide O-linked N-acetyl-D-glucosamine (O-GlcNAc), we evaluated whether increased O-GlcNAcylation affects HIV-1 transcription. In the present study we demonstrate that treatment of HIV-1-infected lymphocytes with the O-GlcNAcylation-enhancing agent glucosamine (GlcN) repressed viral transcription in a dose-dependent manner. Overexpression of O-GlcNAc transferase (OGT), the sole known enzyme catalyzing the addition of O-GlcNAc to proteins, specifically inhibited the activity of the HIV-1 LTR promoter in different T-cell lines and in primary CD4(+) T lymphocytes. Inhibition of HIV-1 LTR activity in infected T cells was most efficient (>95%) when OGT was recombinantly overexpressed prior to infection. O-GlcNAcylation of the transcription factor Sp1 and the presence of Sp1-binding sites in the LTR were found to be crucial for this inhibitory effect. From this study, we conclude that O-GlcNAcylation of Sp1 inhibits the activity of the HIV-1 LTR promoter. Modulation of Sp1 O-GlcNAcylation may play a role in the regulation of HIV-1 latency and activation and links viral replication to the glucose metabolism of the host cell. Hence, the establishment of a metabolic treatment might supplement the repertoire of antiretroviral therapies against AIDS.
Figures
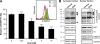
Similar articles
-
HIV-1 transcriptional silencing caused by TRIM22 inhibition of Sp1 binding to the viral promoter.Retrovirology. 2015 Dec 18;12:104. doi: 10.1186/s12977-015-0230-0. Retrovirology. 2015. PMID: 26683615 Free PMC article.
-
Semen Exosomes Promote Transcriptional Silencing of HIV-1 by Disrupting NF-κB/Sp1/Tat Circuitry.J Virol. 2018 Oct 12;92(21):e00731-18. doi: 10.1128/JVI.00731-18. Print 2018 Nov 1. J Virol. 2018. PMID: 30111566 Free PMC article.
-
Hijacking of the O-GlcNAcZYME complex by the HTLV-1 Tax oncoprotein facilitates viral transcription.PLoS Pathog. 2017 Jul 24;13(7):e1006518. doi: 10.1371/journal.ppat.1006518. eCollection 2017 Jul. PLoS Pathog. 2017. PMID: 28742148 Free PMC article.
-
Human immunodeficiency virus type-1 transcription: role of the 5'-untranslated leader region (review).Int J Mol Med. 1998 May;1(5):875-81. doi: 10.3892/ijmm.1.5.875. Int J Mol Med. 1998. PMID: 9852310 Review.
-
A compilation of cellular transcription factor interactions with the HIV-1 LTR promoter.Nucleic Acids Res. 2000 Feb 1;28(3):663-8. doi: 10.1093/nar/28.3.663. Nucleic Acids Res. 2000. PMID: 10637316 Free PMC article. Review.
Cited by
-
Differential transcriptional and functional properties of regulatory T cells in HIV-infected individuals on antiretroviral therapy and long-term non-progressors.Clin Transl Immunology. 2021 May 26;10(5):e1289. doi: 10.1002/cti2.1289. eCollection 2021. Clin Transl Immunology. 2021. PMID: 34094548 Free PMC article.
-
Nutrient regulation of signaling and transcription.J Biol Chem. 2019 Feb 15;294(7):2211-2231. doi: 10.1074/jbc.AW119.003226. Epub 2019 Jan 9. J Biol Chem. 2019. PMID: 30626734 Free PMC article. Review.
-
Cross talk between O-GlcNAcylation and phosphorylation: roles in signaling, transcription, and chronic disease.Annu Rev Biochem. 2011;80:825-58. doi: 10.1146/annurev-biochem-060608-102511. Annu Rev Biochem. 2011. PMID: 21391816 Free PMC article. Review.
-
Small molecules targeted to a non-catalytic "RVxF" binding site of protein phosphatase-1 inhibit HIV-1.PLoS One. 2012;7(6):e39481. doi: 10.1371/journal.pone.0039481. Epub 2012 Jun 29. PLoS One. 2012. PMID: 22768081 Free PMC article.
-
O-GlcNAc modification of transcription factor Sp1 mediates hyperglycemia-induced VEGF-A upregulation in retinal cells.Invest Ophthalmol Vis Sci. 2014 Oct 28;55(12):7862-73. doi: 10.1167/iovs.14-14048. Invest Ophthalmol Vis Sci. 2014. PMID: 25352121 Free PMC article.
References
-
- Blough, H. A., and R. L. Giuntoli. 1979. Successful treatment of human genital herpes infections with 2-deoxy-D-glucose. JAMA 2412798-2801. - PubMed
-
- Bonehill, A., C. Heirman, S. Tuyaerts, A. Michiels, K. Breckpot, F. Brasseur, Y. Zhang, P. Van Der Bruggen, and K. Thielemans. 2004. Messenger RNA-electroporated dendritic cells presenting MAGE-A3 simultaneously in HLA class I and class II molecules. J. Immunol. 1726649-6657. - PubMed
-
- Bonello, M. R., and L. M. Khachigian. 2004. Fibroblast growth factor-2 represses platelet-derived growth factor receptor-alpha (PDGFR-alpha) transcription via ERK1/2-dependent Sp1 phosphorylation and an atypical cis-acting element in the proximal PDGFR-alpha promoter. J. Biol. Chem. 2792377-2382. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

