Correlations of inhibitor kinetics for Pneumocystis jirovecii and human dihydrofolate reductase with structural data for human active site mutant enzyme complexes
- PMID: 19196009
- PMCID: PMC2765523
- DOI: 10.1021/bi801960h
Correlations of inhibitor kinetics for Pneumocystis jirovecii and human dihydrofolate reductase with structural data for human active site mutant enzyme complexes
Abstract
To understand the role of specific active site residues in conferring selective dihydrofolate reductase (DHFR) inhibition from pathogenic organisms such as Pneumocystis carinii (pc) or Pneumocystis jirovecii (pj), the causative agent in AIDS pneumonia, it is necessary to evaluate the role of these residues in the human enzyme. We report the first kinetic parameters for DHFR from pjDHFR and pcDHFR with methotrexate (MTX), trimethoprim (TMP), and its potent analogue, PY957. We also report the mutagenesis and kinetic analysis of active site mutant proteins at positions 35 and 64 of human (h) DHFR and the crystal structure determinations of hDHFR ternary complexes of NADPH and PY957 with the wild-type DHFR enzyme, the single mutant protein, Gln35Lys, and two double mutant proteins, Gln35Ser/Asn64Ser and Gln35Ser/Asn64Phe. These substitutions place into human DHFR amino acids found at those sites in the opportunistic pathogens pcDHFR (Q35K/N64F) and pjDHFR (Q35S/N64S). The K(i) inhibition constant for PY957 showed greatest potency of the compound for the N64F single mutant protein (5.2 nM), followed by wild-type pcDHFR (K(i) 22 nM) and then wild-type hDHFR enzyme (K(i) 230 nM). Structural data reveal significant conformational changes in the binding interactions of PY957 in the hDHFR Q35S/N64F mutant protein complex compared to the other hDHFR mutant protein complexes and the pcDHFR ternary complex. The conformation of PY957 in the wild-type DHFR is similar to that observed for the single mutant protein. These data support the hypothesis that the enhanced selectivity of PY957 for pcDHFR is in part due to the contributions at positions 37 and 69 (pcDHFR numbering). This insight will help in the design of more selective inhibitors that target these opportunistic pathogens.
Figures
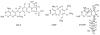



References
-
- Kovacs JA, Gill VJ, Meshnick S, Masur H. New Insights into Transmission, Diagnosis, and Drug Treatment of Pneumocystis carinii Pneumonia. J Amer Med Assoc. 2001;286:2450–2460. - PubMed
-
- Hughes WT. Prevention and Treatment of Pneumocystis carinii Pneumonia. Ann Rev Med. 1991;42:287–295. - PubMed
-
- Totet A, Duwat H, Magois E, Jounieaux V, Roux P, Raccurt C, Nevez G. Similar Genotypes of Pneumocystis jirovecii in Different Forms of Pneumocystis Infection. Microbiology. 2004;150:1173–1178. - PubMed
-
- Benfield T, Atzori C, Miller RF, Helweg-Larsen J. Second-Line Salvage Treatment of AIDS-Associated Pneumocystsis jirovecii Pneumonia. J Acquir Immune Defic Syndr. 2008;48:63–67. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

