Influence of adult mesenchymal stem cells on in vitro vascular formation
- PMID: 19196139
- PMCID: PMC2792097
- DOI: 10.1089/ten.tea.2008.0254
Influence of adult mesenchymal stem cells on in vitro vascular formation
Abstract
The effective delivery of bioactive molecules to wound sites hasten repair. Cellular therapies provide a means for the targeted delivery of a complex, multiple arrays of bioactive factors to wound sites. Thus, the identification of ideal therapeutic populations is an essential aspect of this approach. In vitro assays can provide an important first step toward this goal by selecting populations that are likely suitable for more expensive and time-consuming in vivo assays. In this study, bone marrow-derived mesenchymal stem cells (BM-MSCs) were integrated into a three-dimensional coculture system that supports the development and stabilization of vascular tube-like structures. The presence of a limited number of BM-MSCs resulted in their coalignment with vascular structures, and it further resulted in increased tubule numbers and complexity. Thus, these studies suggest that BM-MSCs functionally interacted with and were attracted to in vitro formed vascular structures. Further, these cells also provided sufficient bioactive factors and matrix molecules to support the formation of tubular arrays and the stabilization of these arrays. This in vitro system provides a means for assessing the function of BM-MSCs in aspects of the angiogenic component of wound repair.
Figures
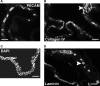

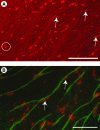





References
-
- Tonnessen M.G. Feng X. Clark R.A.F. Angiogenesis in wound healing. J Investig Dermatol Symp Proc. 2000;5:34. - PubMed
-
- Metcalfe A.D. Ferguson M.W.J. Bioengineering skin using mechanisms of regeneration and repair. Biomaterials. 2007;28:100. - PubMed
-
- Pierce G.F. Tarpley J.E. Tseng J. Bready J. Chang D. Kenney W.C. Rudolph R. Robson M.C. Vande Berge J. Reid P. Kaufman S. Farrell C. Detection of platelet-derived growth factor (PDGF)-AA in actively healing human wounds treated with recombinant PDGF-BB and absence of PDGF in chronic nonhealing wounds. J Clin Invest. 1995;96:1336. - PMC - PubMed
-
- Obara K. Ishihara M. Fujita M. Kanatani Y. Hattori H. Matsui T. Takas B. Ozeki Y. Nakamura S. Ishizuka T. Tominaga S. Hiroi S. Kawai T. Maehara T. Acceleration of wound healing in healing-impaired db/db mice with a photocrosslinkable chitosan hydrogel containing fibroblast growth factor-2. Wound Repair Regen. 2005;13:390. - PubMed
-
- Zymek P. Bujak M. Chatila K. Cieslak A. Thakker G. Entman M.L. Frangogiannis N.G. The role of platelet-derived growth factor signaling in healing myocardial infarcts. J Am Coll Cardiol. 2006;48:2315. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

