Influence of temporary chondroitinase ABC-induced glycosaminoglycan suppression on maturation of tissue-engineered cartilage
- PMID: 19196151
- PMCID: PMC2792113
- DOI: 10.1089/ten.tea.2008.0495
Influence of temporary chondroitinase ABC-induced glycosaminoglycan suppression on maturation of tissue-engineered cartilage
Abstract
Objective: A fundamental challenge of cartilage tissue engineering has been the inability to promote collagen synthesis up to native levels. In contrast, recent protocols have demonstrated that glycosaminoglycans (GAG) can be synthesized to native levels in 4-6 weeks of in vitro culture. We hypothesize that rapid GAG synthesis may be an impediment to collagen synthesis, possibly by altering transport pathways of nutrients or synthesis products. In this study, this hypothesis is tested by inducing enzymatic GAG loss in the early culture period of cartilage tissue constructs, and monitoring collagen content at various time points after cessation of enzymatic treatment.
Methods: In Study 1, to induce breakdown of proteoglycans, chondroitinase ABC (CABC, 0.002U/mL) was continuously added into the culture media for the initial 4 weeks of culture or for 2 weeks starting on day 14 of culture. In Study 2, multiple transient CABC treatments (0.15U/mL, for 2 days) were applied to the matured tissue-engineered constructs.
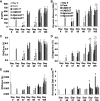
Results: Continuous and transient CABC treatments significantly increased the collagen concentration of the constructs, improving their tensile properties. The GAG content of the treated constructs recovered quickly to the pretreatment level after 2-3 weeks.
Conclusions: This study demonstrates that tissue-engineered cartilage constructs with improved tensile properties can be achieved by temporarily suppressing the GAG content enzymatically.
Figures



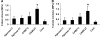

References
-
- Guilak F. Sah R.L. Setton L.A. Physical regulation of cartilage metabolism. In: Mow V.C., editor; Hayes W.C., editor. Basic Orthopaedic Biomechanics. Philadelphia: Lippincott-Raven; 1997. pp. 179–207.
-
- Mow V.C. Bachrach N.M. Setton L.A. Guilak F. Stress, strain, pressure, and flow fields in articular cartilage and chondrocytes. In: Mow V.C., editor; Guilak F., editor; Hayes W.C., editor; Tran-Son-Tay R., editor; Hochmuth R.M., editor. Cell Mechanics and Cellular Engineering. New York: Springer-Verlag; 1994. pp. 345–79.
-
- Asanbaeva A. Masuda K. Thonar E.J. Klisch S.M. Sah R.L. Mechanisms of cartilage growth: modulation of balance between proteoglycan and collagen in vitro using chondroitinase ABC. Arthritis Rheum. 2007;56:188. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

