Aging impairs skeletal muscle mitochondrial bioenergetic function
- PMID: 19196905
- PMCID: PMC2691197
- DOI: 10.1093/gerona/gln048
Aging impairs skeletal muscle mitochondrial bioenergetic function
Abstract
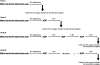
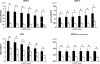
This study investigated the influence of age on the functional status of mitochondria isolated from skeletal muscle of C57BL/6 mice aged 3 and 18 months. We hypothesized that skeletal muscle mitochondria isolated from aged animals will exhibit a decreased respiratory function. Mitochondrial respiratory functional measures (ie, State 3 and 4 respiration, respiratory control ratio and number of nanomoles of ADP phosphorylated by nanomoles of O(2) consumed per mitochondrion) and biochemical markers of oxidative damage (aconitase activity, protein carbonyl derivatives, sulfhydryl groups, and malondialdehyde) were measured in isolated mitochondrial suspensions. Along with traditional tests of mitochondrial function, an in vitro repetitive ADP-stimulation test was used to evaluate the mitochondrial capacity to reestablish the homeostatic balance between successive ADP stimulations. The number of mitochondria per mitochondrial suspension, calculated by transmission electron microscopy, was used to normalize functional and biochemical data. Our results confirm the existence of an age-associated decline in mitochondrial function of mixed skeletal muscle, which is significantly correlated with higher levels of mitochondrial oxidative damage.
Figures

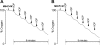
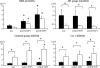
References
-
- Nicholls DG. Mitochondrial function and dysfunction in the cell: its relevance to aging and aging-related disease. Int J Biochem Cell Biol. 2002;34:1372–1381. - PubMed
-
- Kwong LK, Sohal RS. Age-related changes in activities of mitochondrial electron transport complexes in various tissues of the mouse. Arch Biochem Biophys. 2000;373::16–22. - PubMed
-
- Salvioli S, Bonafe M, Capri M, Monti D, Franceschi C. Mitochondria, aging and longevity—a new perspective. FEBS Lett. 2001;492:9–13. - PubMed
-
- Cadenas E, Davies KJ. Mitochondrial free radical generation, oxidative stress, and aging. Free Radic Biol Med. 2000;29:222–230. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

