Oncogenic EGFR signaling cooperates with loss of tumor suppressor gene functions in gliomagenesis
- PMID: 19196966
- PMCID: PMC2650331
- DOI: 10.1073/pnas.0813314106
Oncogenic EGFR signaling cooperates with loss of tumor suppressor gene functions in gliomagenesis
Abstract
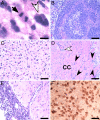
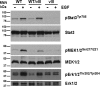
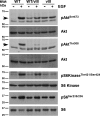
Glioblastoma multiforme (GBM) is a highly lethal brain tumor for which little treatment is available. The epidermal growth factor receptor (EGFR) signaling pathway is thought to play a crucial role in GBM pathogenesis, initiating the early stages of tumor development, sustaining tumor growth, promoting infiltration, and mediating resistance to therapy. The importance of this pathway is highlighted in the fact that EGFR is mutationally activated in over 50% of GBM tumors. Consistent with this, we show here that concomitant activation of wild-type and/or mutant (vIII) EGFR and ablation of Ink4A/Arf and PTEN tumor suppressor gene function in the adult mouse central nervous system generates a fully penetrant, rapid-onset high-grade malignant glioma phenotype with prominent pathological and molecular resemblance to GBM in humans. Studies of the activation of signaling events in these GBM tumor cells revealed notable differences between wild-type and vIII EGFR-expressing cells. We show that wild-type EGF receptor signals through its canonical pathways, whereas tumors arising from expression of mutant EGFR(vIII) do not use these same pathways. Our findings provide critical insights into the role of mutant EGFR signaling function in GBM tumor biology and set the stage for testing of targeted therapeutic agents in the preclinical models described herein.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Kleihues P, Cavenee WK. Pathology and Genetics of Tumours of the Nervous System. Lyon, France: International Agency for Research on Cancer; 2000.
-
- Pedersen MW, Meltorn M, Damstrup L, Poulsen HS. The type III epidermal growth factor receptor mutation. Biological significance and potential target for anti-cancer therapy. Ann Oncol. 2001;12:745–760. - PubMed
-
- Ekstrand AJ, et al. Genes for epidermal growth factor receptor, transforming growth factor alpha, and epidermal growth factor and their expression in human gliomas in vivo. Cancer Res. 1991;51:2164–2172. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

