Cascade reactions during coronafacic acid biosynthesis: elongation, cyclization, and functionalization during Cfa7-catalyzed condensation
- PMID: 19199623
- PMCID: PMC2662718
- DOI: 10.1021/ja8077945
Cascade reactions during coronafacic acid biosynthesis: elongation, cyclization, and functionalization during Cfa7-catalyzed condensation
Abstract
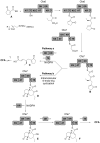
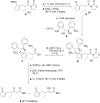
Herein, the biogenesis of the hydrindane ring system within coronafacic acid (CFA) has been investigated. These studies reveal that in addition to the canonical polyketide chain elongation and functionalization encoded by type I polyketide synthase (PKSs), cascade reactions can take place during assembly line-like biosynthesis. Indeed, upon Cfa7-catalyzed Claisen condensation between enzyme-bound malonate and an N-acetylcysteamine (SNAC) thioester, latent reactivity within the elongated enzyme-bound intermediate is unveiled. This reactivity translates into an intramolecular cyclization, which can proceed in a facile manner as observed by the enzyme-independent cyclization of a linear beta-ketothioester intermediate.
Figures
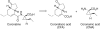



Similar articles
-
Analysis of the enzymatic domains in the modular portion of the coronafacic acid polyketide synthase.Gene. 2001 May 30;270(1-2):191-200. doi: 10.1016/s0378-1119(01)00476-0. Gene. 2001. PMID: 11404016
-
Structure and function of an iterative polyketide synthase thioesterase domain catalyzing Claisen cyclization in aflatoxin biosynthesis.Proc Natl Acad Sci U S A. 2010 Apr 6;107(14):6246-51. doi: 10.1073/pnas.0913531107. Epub 2010 Mar 23. Proc Natl Acad Sci U S A. 2010. PMID: 20332208 Free PMC article.
-
Biosynthesis of the Pseudomonas polyketide coronafacic acid requires monofunctional and multifunctional polyketide synthase proteins.Proc Natl Acad Sci U S A. 1998 Dec 22;95(26):15469-74. doi: 10.1073/pnas.95.26.15469. Proc Natl Acad Sci U S A. 1998. PMID: 9860992 Free PMC article.
-
Biosynthesis of biphenyls and benzophenones--evolution of benzoic acid-specific type III polyketide synthases in plants.Phytochemistry. 2009 Oct-Nov;70(15-16):1719-27. doi: 10.1016/j.phytochem.2009.06.017. Epub 2009 Aug 21. Phytochemistry. 2009. PMID: 19699497 Review.
-
Biomimetic Thioesters as Probes for Enzymatic Assembly Lines: Synthesis, Applications, and Challenges.Cell Chem Biol. 2016 Oct 20;23(10):1179-1192. doi: 10.1016/j.chembiol.2016.08.014. Epub 2016 Sep 29. Cell Chem Biol. 2016. PMID: 27693058 Review.
Cited by
-
Nonproteinogenic amino acid building blocks for nonribosomal peptide and hybrid polyketide scaffolds.Angew Chem Int Ed Engl. 2013 Jul 8;52(28):7098-124. doi: 10.1002/anie.201208344. Epub 2013 May 31. Angew Chem Int Ed Engl. 2013. PMID: 23729217 Free PMC article. Review.
-
Discovery, characterization and engineering of ligases for amide synthesis.Nature. 2021 May;593(7859):391-398. doi: 10.1038/s41586-021-03447-w. Epub 2021 May 19. Nature. 2021. PMID: 34012085
-
Cyclization of aromatic polyketides from bacteria and fungi.Nat Prod Rep. 2010 Jun;27(6):839-68. doi: 10.1039/b911518h. Epub 2010 Mar 31. Nat Prod Rep. 2010. PMID: 20358042 Free PMC article. Review. No abstract available.
-
Structural insights into nonribosomal peptide enzymatic assembly lines.Nat Prod Rep. 2009 Aug;26(8):987-1000. doi: 10.1039/b904543k. Epub 2009 May 22. Nat Prod Rep. 2009. PMID: 19636447 Free PMC article. Review.
-
Acyltransferase mediated polyketide release from a fungal megasynthase.J Am Chem Soc. 2009 Jun 24;131(24):8388-9. doi: 10.1021/ja903203g. J Am Chem Soc. 2009. PMID: 19530726 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

